A Generalization of the Ternary Binding Model to Membrane-Confined Systems with Finite Copy Number
A Generalization of the Ternary Binding Model to Membrane-Confined Systems with Finite Copy Number
Bellout, H.; Li, A.; Piatkov, K.; Bottino, D.
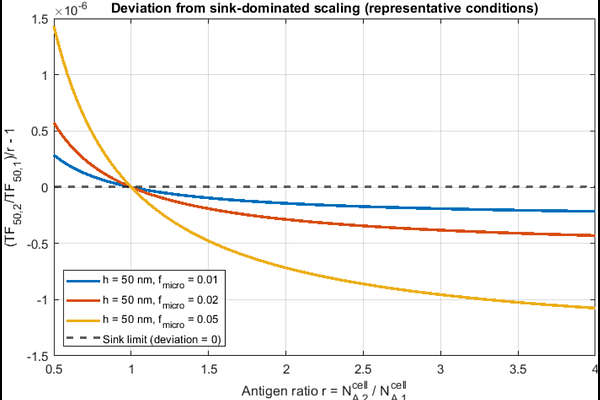
AbstractBispecific T cell engagers and related immunotherapies are dosed using equilibrium binding models derived for well-mixed solution, yet therapeutic activity occurs at nanoscale membrane synapses with finite receptor copy numbers. Here we show that membrane confinement introduces geometry-dependent corrections to the landmark Douglass [6] ternary binding model, shifting formation half-points (TF50) by 2 10 fold at clinically relevant antigen densities. We present two complementary formulations effective concentration and surface density that preserve the Douglass framework while explicitly accounting for synapse geometry, surface topology, and the accessibility factor of surface receptors. We further derive stochastic descriptions of trimer formation via the chemical master equation, demonstrating the recovery of the classical Ternary Binding Model equilibrium in appropriate limits. We illustrate the framework using the CD19-targeting BiTE blinatumomab as a case study. Accounting for microvillus-driven patchy close contact during immune surveillance yields a mechanistic explanation for why higher target antigen density can increase the dose required to achieve a fixed level of ternary formation: in the membrane-confined regime, excess target acts as a local antigen sink that sequesters drug and reduces the free fraction available for productive bridging. Rather than fitting to a single shift value, we emphasize the robust scaling and regime structure predicted by the theory (density-proportional behavior in the sink-dominated limit, and collapse toward affinity-limited behavior outside that limit). The generalized framework provides ready-to-use correction formulas and parameter-estimation guidance, establishing a rigorous foundation for antigen-density-aware dosing strategies in T cell engager pharmacology.