Loss of enteric BDNF TrkB signaling and VIPergic dysfunction underlie gastrointestinal dysmotility in a Mecp2-null mouse model of Rett syndrome
Loss of enteric BDNF TrkB signaling and VIPergic dysfunction underlie gastrointestinal dysmotility in a Mecp2-null mouse model of Rett syndrome
Puttapaka, S. N.; Admasu, I. A.; Scott, A.; Sonmez, G.; Seika, P.; Rajkumar, M.; Valencia, X.; Consorti, A.; Hong, S. M.; Slosberg, J.; Fagiolini, M.; Kulkarni, S.
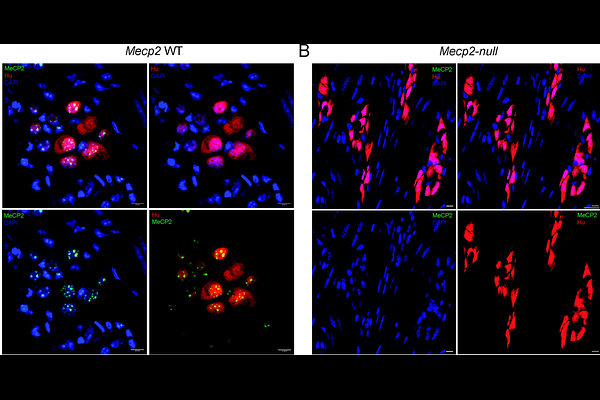
AbstractGastrointestinal (GI) dysmotility is a highly prevalent and clinically significant feature of Rett syndrome (RTT), yet its underlying mechanisms remain poorly defined. Here, we investigated these mechanisms of GI dysmotility in a Mecp2null mouse model of RTT. First, we observed that MeCP2 was expressed in murine myenteric ganglia, including in enteric neurons and that Mecp2null males developed maturation associated functional regression in their GI motility. In dysmotile mice, longitudinal muscle myenteric plexus tissue showed marked reductions in enteric Bdnf isoforms IV, VI, and II, whereas expression of the BDNF receptor isoforms TrkB.FL and TrkB.T1 was not significantly altered, consistent with reduced enteric BDNF TrkB signaling. Despite impaired GI motility, Mecp2null mice showed no significant changes in total enteric neuronal density, nitrergic neuronal abundance, or expression of Nos1, Chat, and Uchl1. In contrast, Vip expression was significantly reduced, while expression of VIP receptor genes: Vipr1 and Vipr2 was increased, indicating disrupted VIPergic signaling. Integration with publicly available enteric single-cell/nucleus datasets and targeted qRTPCR further suggested altered inhibitory neuronal subtype composition, with reduced Vip+ Cartpt+ signatures and increased Nfia expression, suggesting that MeCP2 loss differentially affects distinct inhibitory neuronal subpopulations. Finally, conditional loss of TrkB.FL in neural crest-derived cells reduced Vip expression without recapitulating the full Mecp2null VIPergic phenotype, indicating that impaired BDNF TrkB signaling contributes to, but does not completely explain, the GI dysmotility in this model of RTT. Together, these findings identify enteric BDNF TrkB and VIPergic dysfunction as key mechanisms underlying GI dysmotility in RTT.