Expanded stoichiometric model of chondrocyte metabolism: response to cyclical shear and compressive loading
Expanded stoichiometric model of chondrocyte metabolism: response to cyclical shear and compressive loading
Kimmel, A.; Arnold, A. D.; Erdogan, A. E.; Komminni, R.; Myers, E.; Cummins, B.; Carlson, R.; June, R.
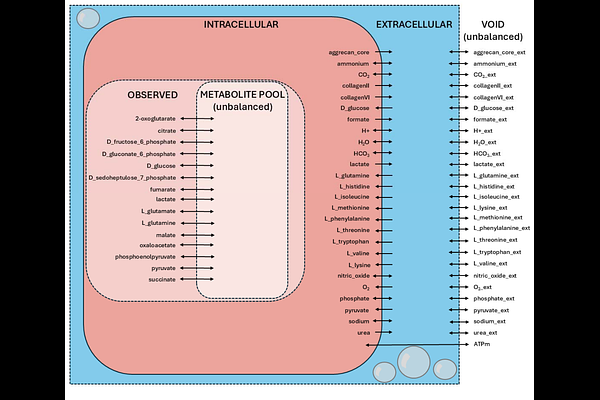
AbstractCartilage deterioration is a hallmark of the most common joint disease, osteoarthritis, and there is substantial interest in developing strategies for cartilage synthesis and repair. Cyclical mechanical stimulation has been known for decades to drive synthesis of cartilage matrix proteins. Matrix synthesis requires activation of central metabolism for producing precursors to non-essential amino acids required for protein translation. However, there are gaps in knowledge regarding how mechanical stimuli affect chondrocyte central metabolism. Here, we find that cyclical shear and compression drive differences in chondrocyte central metabolism in a sex-dependent manner. Based on established biochemistry, we developed and validated a stoichiometric model containing 139 metabolites and 172 reactions from central metabolism that includes production of the key cartilage matrix proteins of types II and VI collagen and aggrecan. We then used experimental data from shear and compressive stimulation of osteoarthritis chondrocytes to constrain this model and ran multiple simulations examining flux through the production reactions of matrix proteins and ATP. Our results show that both shear and compression can stimulate osteoarthritic chondrocyte metabolism in a manner consistent with production of cartilage matrix proteins, with notable differences in simulated central metabolism between male and female chondrocytes. Additionally, and importantly, our simulation results suggest that nitrogen availability is a key limitation to chondrocyte synthesis of matrix proteins. These results are a starting point for using central metabolism of chondrocytes to optimize synthesis of matrix proteins for cartilage repair. For example, increasing glutamine levels in the presence of cyclical compression has potential to increase production of both types II and VI collagen. These strategies have potential for improving cartilage tissue engineering and repair.