Emergence of rigidity percolation and critical behavior in tunable protein condensates
Emergence of rigidity percolation and critical behavior in tunable protein condensates
Liao, Z.; Jia, B.; Xu, Y.; Shen, Z.; Zhang, M.; Tong, P.
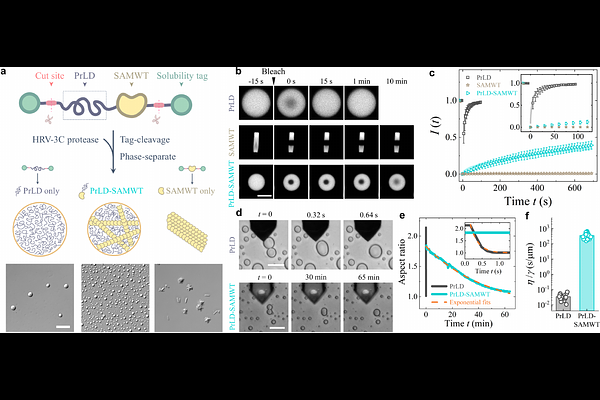
AbstractMultivalent proteins are known to form complex networks within biomolecular condensates, yet the mechanisms governing the emergence and evolution of these networks remain poorly understood. Here, we utilize a synthetic protein chimera system with tunable interactions to investigate the transition from liquid-like droplets to networked condensates. By employing single amino-acid substitutions, we generated a series of mutants with varying protein-binding strengths. As the interaction strength increases, the condensates undergo a sharp rigidity percolation transition, characterized by a more than 200-fold increase in both elastic modulus and viscosity. Near this transition, we identify a critical scaling relation in the condensate elasticity, providing robust evidence for a percolation-driven assembly mechanism. Furthermore, we demonstrate that this network architecture is fundamentally linked to biological functions. The introduction of disease associated mutations disrupts the network, significantly softening the condensates and rendering them fluid-like. Our findings reveal that network percolation and criticality in protein condensates can be sensitively regulated by single-amino-acid substitutions, underscoring their essential role in maintaining structural integrity and supporting physiological function.