Divergent successional patterns and infection dynamics in virion and transcriptionally active soil viral communities following phosphorus amendment and wet-up
Divergent successional patterns and infection dynamics in virion and transcriptionally active soil viral communities following phosphorus amendment and wet-up
Gogul, G.; Allen, G. M.; Leleiwi, I.; Blazewicz, S. J.; Pett-Ridge, J.; Emerson, J. B.; Trubl, G.
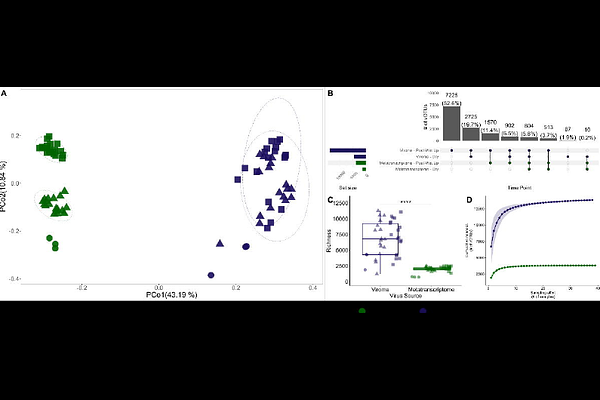
AbstractViruses are key regulators of terrestrial carbon, nitrogen, and phosphorus cycling, yet how environmental perturbations structure viral activity remains poorly resolved. Rewetting of seasonally dry soils triggers rapid microbial and viral responses, but the relationships between virion-associated and transcriptionally active viral communities, and the role of phosphorus in these dynamics, remain unclear. Here, we integrated viromics, metatranscriptomics, environmental DNA (eDNA), and amplicon sequencing to track viral succession and virus-host interactions over three weeks following soil rewetting, with and without phosphorus amendment. We identified 13,840 viral populations (vOTUs), of which 3,803 were transcriptionally active, representing ongoing infections. Wet-up significantly altered virion and transcriptionally active viral communities, while phosphorus selectively influenced prokaryotic and transcriptionally active viral communities but not virion composition. Virus-host linkages were predicted for 32% of vOTUs, with transcriptionally active bacteriophages infecting Actinomycetota increasing under phosphorus amendment. Following wet-up, virion abundance decreased ~3-fold while virocells increased ~5-fold, indicating a shift from viral persistence in dry soils to active infection. Phosphorus further enhanced virocell abundance. eDNA captured rapid viral turnover and revealed transient dynamics not resolved by viromes or metatranscriptomes alone. Together, these results demonstrate that soil viral communities are structured by distinct but complementary molecular pools that operate over different ecological timescales. Wet-up activates a reservoir of persistent virions, while phosphorus availability regulates infection dynamics and host-virus coupling. These findings highlight viruses as dynamic drivers of microbial turnover and nutrient cycling following environmental perturbation, advancing a more predictive understanding of soil ecosystem responses to changing resource availability.