Distinct postsynaptic morphogenetic strategies across Drosophila embryonic muscles during neuromuscular junction formation
Distinct postsynaptic morphogenetic strategies across Drosophila embryonic muscles during neuromuscular junction formation
Inal, M. A.; Kamiyama, D.
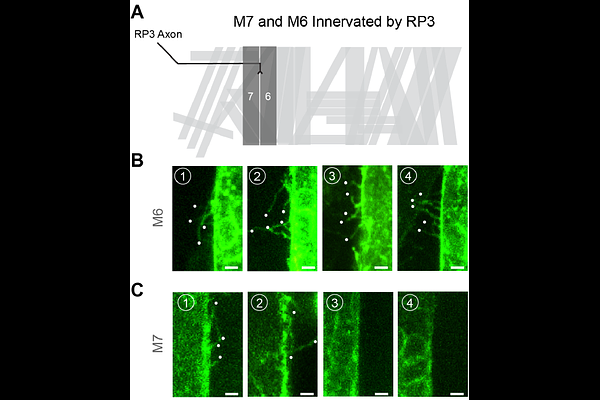
AbstractPrecise synaptic connectivity emerges through coordinated interactions between neurons and their target cells during development. At the Drosophila embryonic neuromuscular junction (NMJ), postsynaptic muscle fibers actively participate in this process by extending dynamic, actin-rich protrusions termed myopodia that interact with approaching motor growth cones. Previous work focusing on muscle 12 (M12) revealed that myopodia cluster at nascent neuron-muscle contact sites, suggesting that specialized postsynaptic architectures may facilitate synaptic partner selection. However, whether similar morphogenetic strategies operate across the diverse set of embryonic muscles has remained unclear. Here, we establish a genetic imaging toolkit that enables minimally invasive visualization of defined muscle subsets throughout the embryo. Using muscle-specific and stochastic GAL4 drivers to label muscle membranes in vivo, we systematically compare myopodial organization across multiple muscle fibers, including M12, M14, M6, and M7. We find that postsynaptic morphology varies substantially between muscles. M12 displays robust myopodial clustering associated with a prominent sheet-like membrane structure, which we term the muscle lamella, whereas M6 and M14 frequently form myopodial clusters but do not evidently exhibit this structure. In contrast, M7 shows markedly reduced clustering frequency and smaller clusters. These observations reveal previously unrecognized heterogeneity in postsynaptic organization among neighboring muscles during early neuromuscular development. Together, our findings demonstrate that myopodial clustering represents a broadly deployed but differentially organized strategy by which muscles engage motor axons during synaptic partner selection. The imaging toolkit established here provides a foundation for systematic analysis of neuron-muscle interactions across the embryonic musculature and reveals that distinct muscles employ diverse morphogenetic strategies during NMJ assembly.