Vorinostat Rescues SQSTM1 Palmitoylation and Restores Dysfunctional Autophagy in Huntington Disease
Vorinostat Rescues SQSTM1 Palmitoylation and Restores Dysfunctional Autophagy in Huntington Disease
Alshehabi, Y.; Abrar, F.; Rabu, M.; Dang, A.; Ramzan, F.; Martin, D. D. O.
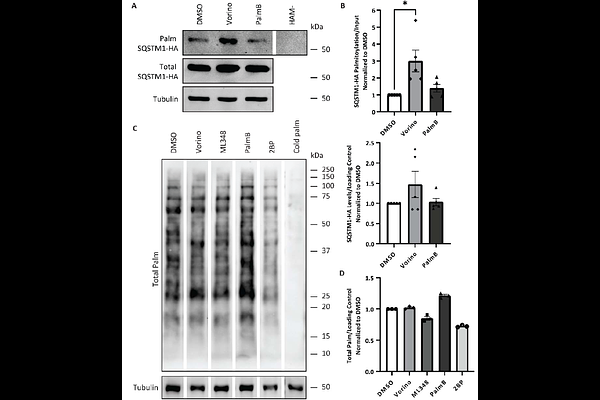
AbstractProtein mislocalization, impaired autophagy, and protein aggregation are central features of Huntington disease (HD), yet therapeutic strategies that simply stimulate autophagy risk exacerbating dysfunction in an already compromised pathway. We previously demonstrated that palmitoylation of the autophagy receptor SQSTM1/p62 is reduced in HD patient brains and YAC128 mice, contributing to defective cargo loading and accumulation of mutant huntingtin (mHTT) aggregates. Here, we identify the FDA-approved histone deacetylase inhibitor Vorinostat (also known as suberoylanilide hydroxamic acid; SAHA) as a small molecule that restores SQSTM1 palmitoylation and autophagic function in HD models. Using acyl-biotin exchange and click chemistry assays, we show that Vorinostat selectively increases SQSTM1 palmitoylation without altering total protein palmitoylation. In turn, Vorinostat enhances autophagic flux, decreases SQSTM1 levels, increases LC3-II, and promotes colocalization of SQSTM1, mHTT, and lysosomes, indicating improved cargo delivery and degradation. Importantly, systemic administration of Vorinostat crosses the blood-brain barrier and restores SQSTM1 palmitoylation in YAC128 mouse cortices. Mechanistically, Vorinostat may function through dual pathways: partial inhibition of depalmitoylating enzymes and transcriptional regulation of palmitoylation machinery. These findings build on our identification of SQSTM1 palmitoylation as a therapeutic target for HD and repositions Vorinostat as a candidate modifier of autophagy dysfunction and palmitoylation impairment in HD.