Granulin loss and TMEM106B risk converge on lysosomal C-terminal fragment pathology in frontotemporal dementia
Granulin loss and TMEM106B risk converge on lysosomal C-terminal fragment pathology in frontotemporal dementia
Zeng, Y.; Xiong, J.; Lovchykova, A.; Song, A.; Gitler, S. W.; Abu-Remaileh, M.; Gitler, A. D.
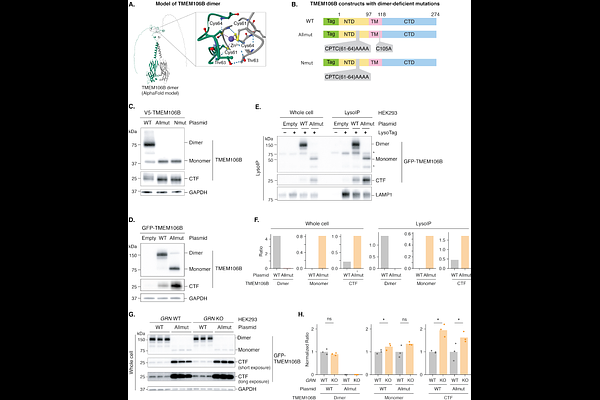
AbstractFrontotemporal dementia (FTD) is the second most common cause of dementia after Alzheimer disease. Mutations in GRN, which encodes progranulin, are a major cause of FTD. Common genetic variants in the TMEM106B gene modify risk of FTD and the effect is especially strong in GRN mutation carriers. Intriguingly, in GRN mutation carriers, being homozygous for the protective TMEM106B haplotype seems to confer near lifetime protection against FTD. Despite the strong genetic link between GRN and TMEM106B, how these two genes interact mechanistically has remained unresolved. Recent studies have revealed that a C-terminal fragment of TMEM106B forms amyloid fibrils and accumulates in the brains of older individuals and patients with neurodegenerative disorders, including FTD. How the production of this fragment connects to granulin deficiency is also unknown. Using lysosome immunoprecipitation, we show that granulin deficiency drives the accumulation of the TMEM106B C-terminal fragment within lysosomes in Grn-knockout mice and GRN-null human iPSC-derived neurons. Recombinant progranulin supplementation reduced TMEM106B C-terminal fragment accumulation. Isogenic neurons carrying the TMEM106B risk allele displayed allele-dose-dependent fragment accumulation that was reversible by progranulin. Structural and genetic analyses demonstrated that TMEM106B dimerization stabilizes the protein and limits C-terminal fragment formation. These findings define a lysosomal pathway linking granulin deficiency to TMEM106B C-terminal fragment accumulation and explain how protective TMEM106B alleles can confer resistance to FTD, even for GRN mutation carriers.