Hydration and hydrolysis define antibiotic resistance conferred by macrolide esterases
Hydration and hydrolysis define antibiotic resistance conferred by macrolide esterases
Kelly, E. T. R.; Myziuk, I.; Hemmings, M. Z.; Mulla, Z.; Blanchet, J.; Ruzzini, A.; Berghuis, A. M.
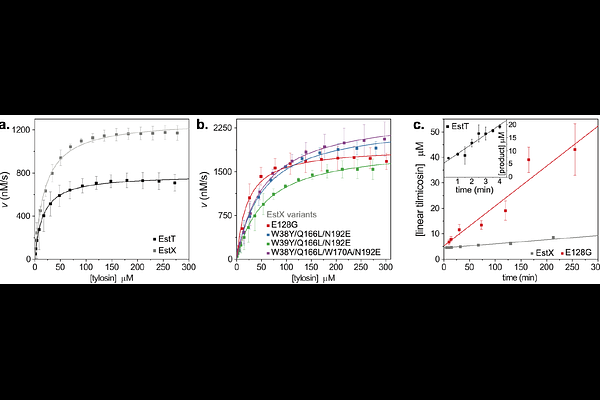
AbstractMacrolides are an antibiotic class widely used in both human and veterinary medicine, and function by interfering with protein synthesis. Regrettably, numerous strategies for evading the antibiotic properties of macrolides have been found in bacteria, including enzyme-mediated inactivation. These mechanisms are now widely disseminated among pathogenic, animal-associated and environmental bacteria making them a One Health issue. Macrolide esterases, which hydrolyze the macrolactones ester bond, confer one such resistance mechanism. Two types of macrolide esterases have thus far been identified, the well-studied erythromycin esterases and the recently discovered Est-type enzymes that belong to the /{beta}-hydrolase superfamily. We present detailed structure-function studies for four diverse Est type esterases: which only share 44-66% sequence identity (EstTSf, EstTSt, EstTBc, and EstXEc). In addition to resistance profiling and substrate specificity studies, we present structures for all four enzymes, including structures for EstTBc and EstXEc in complex with tylosin and tylvalosin macrolides, post hydrolysis. Complementing the data with mutational and kinetic studies allowed for a detailed analysis of the structural basis for macrolide-enzyme interactions. Combined the data suggest that promiscuous binding and imprecise positioning, mediated by a water-cage, dictate substrate specificity for Est-type macrolide resistance enzymes. These insights may prove beneficial for next-generation antibiotic development.