c-di-AMP inactivates a K+/H+ antiporter in Bacillus subtilis
c-di-AMP inactivates a K+/H+ antiporter in Bacillus subtilis
Figueiredo-Costa, I. R.; Lorga-Gomes, M. M.; Sousa-Moreira, S. C.; Matas, I. M.; Morais-Cabral, J. H.
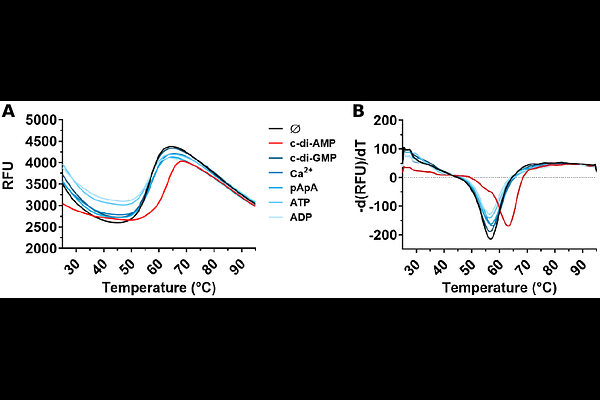
Abstractc-di-AMP is a bacterial second messenger with the crucial role of regulating turgor and osmotic adaptation. Due to the importance of intracellular K+ for osmotic balance, c-di-AMP controls the import and export of K+ by regulating the activity and transcription level of K+ transporters and channels. It has been postulated that c-di-AMP inactivates K+ import and activates K+ export. To gain a full understanding of the properties the K+ machinery in the Gram-positive model organism Bacillus subtilis and in particular, of how the machinery is regulated by c-di-AMP, we characterized the molecular properties of CpaA, a cation/H+ antiporter that has been shown to bind the dinucleotide. We determined the crystal structure of the cytosolic RCK domain with and without c-di-AMP and performed a functional characterization of full-length CpaA using a fluorescence-based flux assay. We found that c-di-AMP binds on the interface of the RCK-C subdomain but only small structural differences are detected between the apo- and holo-structure. We determined that CpaA is more active at high pH and that it slightly favors K+ over Na+ for exchange with H+. Unexpectedly, CpaA is inactivated by c-di-AMP with a K1/2 of inactivation around 1 M. Our results reinforce the emerging view that regulation of the bacterial K+ machinery by c-di-AMP is more complex than previously thought and that a detailed characterization of the molecular properties of the individual protein components and of how their activity is integrated is necessary for a complete view of the machinery physiological function.