Structure of human aldehyde oxidase under tris(2-carboxyethyl)phosphine-reducing conditions
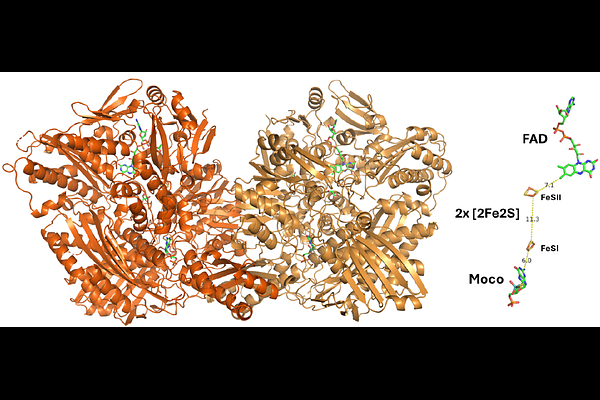
Structure of human aldehyde oxidase under tris(2-carboxyethyl)phosphine-reducing conditions
Videira, C.; Esmaeeli, M.; Leimkuhler, S.; Romao, M. J.; Mota, C.
AbstractThe importance of human aldehyde oxidase (hAOX1) has increased over the last decades due to its involvement in drug metabolism. Inhibition studies concerning hAOX1 are extensive and a common reducing agent, dithiothreitol (DTT), was recently found to inactivate the enzyme. However, in previous crystallographic studies of hAOX1, DTT was found to be essential for crystallization. To surpass this concern another reducing agent used in crystallization trials. Using tris(2-carboxyethyl)phosphine (TCEP), a sulphur-free reducing agent, it was possible to obtain well-ordered crystals from hAOX1 wild type and variant, hAOX1_6A, which diffracted beyond 2.3 A. Instead of the typical star-shaped crystals of hAOX1, at pH 4.7, plates are obtained in the orthorhombic space group (P22121) with two molecules in the asymmetric unit. Activity assays with the enzyme incubated with both reducing agents show that contrary to DTT, TCEP does not lead to irreversible inactivation of the enzyme. The replacement of DTT with TCEP in crystallization of hAOX1 provides a strategy to circumvent enzyme inactivation during crystallographic studies, allowing future applications of new assays, such as time-resolved crystallography.