Genomic insights into polyketide toxin synthesis and algal symbiosis using high-quality genome sequences of the early divergent hexacorallian genus Palythoa (Cnidaria, Zoantharia)
Genomic insights into polyketide toxin synthesis and algal symbiosis using high-quality genome sequences of the early divergent hexacorallian genus Palythoa (Cnidaria, Zoantharia)
Yoshioka, Y.; Shoguchi, E.; Chiu, Y.-L.; Kawamitsu, M.; Reimer, J. D.; Yamashita, H.
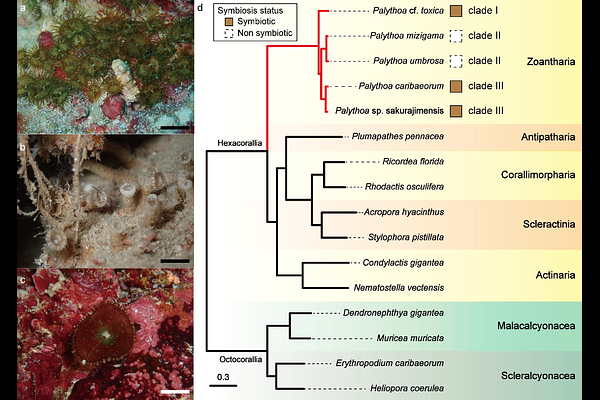
AbstractPalytoxin, first isolated from Palythoa toxica, is among the most potent marine toxins known. Despite decades of biochemical investigation, genetic bases underlying its potential biosynthesis in Palythoa remain unresolved. Here we present four high-quality genome assemblies of Palythoa species, including Palythoa cf. toxica, and integrate these with a chromosome-scale genome assembly of P. caribaeorum. Performing comparative genomic analyses, we screened for candidate genes potentially involved in palytoxin biosynthesis and examined patterns of genome evolution. Unexpectedly, we identified only two classes of ketosynthase (KS) domain-containing genes in Palythoa: fatty acid synthases (FAS) and bacterial-like polyketide synthases (PKSs). Contrasting other anthozoans, animal FAS-like PKS (AFPK) genes common to all Palythoa species were not detected. We found no evidence for lineage-specific expansion of PKS genes unique to Palythoa, suggesting that if palytoxin/palytoxin-like molecule biosynthesis is host-encoded, it may involve functional modification or co-opting pre-existing FAS and/or bacterial-like PKS pathways. Comparative analyses revealed expansions of gene families associated with transport and binding functions in Palythoa, potentially reflecting molecular adaptations linked to their sand-incorporating body structure. We identified TPT1 and CLEC4A as rapidly evolving genes in multiple Palythoa species, consistent with possible roles in growth regulation and host-microbe interactions. Additionally, comparison between azooxanthellate and zooxanthellate species revealed mutations within conserved protein domains of LePin, which has been implicated in cnidarian endosymbiosis, suggesting lineage-specific modifications associated with symbiotic state. This study establishes a foundation for zoantharian genomic research, provides insights into lineage-specific genomic signatures, and advances molecular and evolutionary biological knowledge of this ecologically important group.