Dengue virus NS1 undergoes partial nuclear translocation to modulate host transcription and support viral replication
Dengue virus NS1 undergoes partial nuclear translocation to modulate host transcription and support viral replication
Pacheco, C. A.; Cruz, R.; Wood, C. D.; Zusinaite, E.; Merits, A.; Avila-Bonilla, R. G.; Garcia-Villegas, R.; Ludert, J. E.
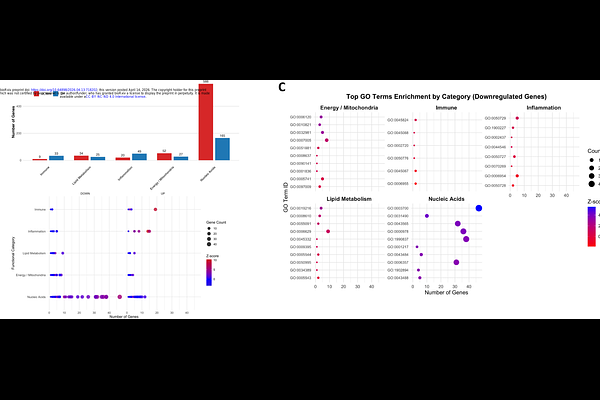
AbstractThe dengue virus (DENV) non-structural protein 1 (NS1) is a glycoprotein highly conserved among mosquito-borne orthoflaviviruses. NS1 is typically localized in the lumen of the endoplasmic reticulum, where it forms part of the replication complexes, and is also exposed at the plasma membrane. In addition, NS1 is secreted as a lipoprotein. Here, using a combination of approaches, including confocal microscopy with deconvolution, in situ analysis, and biochemical cell fractionation, we show that a substantial fraction of NS1 (up to 30%) translocates to the nucleus during infection. We identified a conserved, structurally exposed bipartite nuclear localization signal (NLS) within NS1. Pharmacological inhibition with ivermectin and site-directed mutagenesis of the NLS in recombinant confirmed that nuclear import of NS1 is an active process, dependent on the classical importin /{beta} pathway. Notably, both dimeric and multimeric forms of NS1 were detected in the nucleus in association with nuclear lamin. Introduction of the NLS mutations into DENV2 infectious clones resulted in a non-viable virus. Production of virus progeny and completion of the replicative cycle by the mutant genomes could be rescued by trans-complementation with wild-type NS1, but not with an NLS-mutated NS1, indicating that an NS1 nuclear phase is required for a productive infection. Transcriptomic analysis by RNA-seq further revealed that NS1 functions depend on its subcellular location. Nuclear NS1 induced the overexpression of genes associated with DNA- binding transcription factors, whereas NLS-mutated NS1, retained in the cytoplasm, failed to induce these genes and instead triggered pro-inflammatory and metabolic responses. Together, these findings reveal a previously unrecognized nuclear phase of NS1 that is required for an efficient viral life cycle, redefining NS1 as a modulator of the host transcriptional environment. These findings also suggest new avenues for antiviral and vaccine development.