Cell divisions both challenge and refine tissue boundaries in the Drosophila embryo
Cell divisions both challenge and refine tissue boundaries in the Drosophila embryo
Castle, V.; Miles, M.; Fernandez-Gonzalez, R.; Erdemci-Tandogan, G.
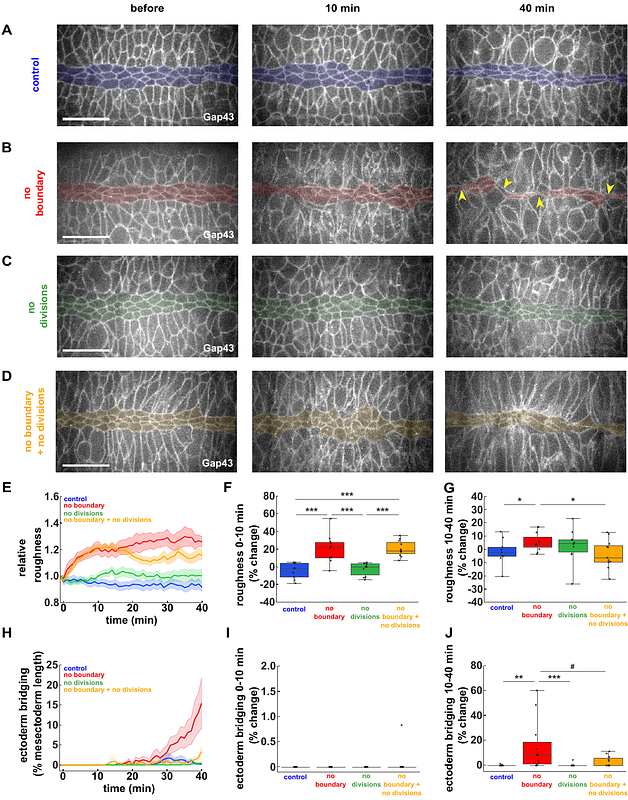
AbstractTissue boundaries pattern embryos, suppress tumours, and provide directional cues. Tissue boundaries are associated with supracellular cables formed by actin and the molecular motor non-muscle myosin II. Actomyosin cables generate tension that prevents cell mixing. Whether other cellular behaviours contribute to the formation of linear interfaces between cell populations remains unclear. In the Drosophila embryo, an actomyosin-based boundary separates the ectoderm from the mesectoderm, a group of neuronal and glial progenitors. Mathematical modelling predicted that cell divisions in the ectoderm challenge the mesectoderm-ectoderm (ME) boundary. Consistent with this, suppressing ectoderm cell divisions in vivo prevented cell mixing across the ME boundary when actomyosin-based tension was lost. Our mathematical model also predicted that cell divisions sharpen the ME boundary by reducing tension and increasing cell motility in the ectoderm. We found that inhibiting ectoderm divisions in vivo reduced boundary linearity. Using laser ablation and cell tracking, we demonstrated that cell divisions reduced junctional tension and increased cell movement in the ectoderm. Together, our results reveal that cell divisions facilitate cellular rearrangements to increase fluidity in a novel mechanism for boundary refinement.