Proteomic profiling of whole tissue sections in cardiac ATTR amyloidosis reveals increased extracellular matrix remodeling
Proteomic profiling of whole tissue sections in cardiac ATTR amyloidosis reveals increased extracellular matrix remodeling
Vandendriessche, A.; Maia, T. M.; Timmermans, F.; Van Haver, D.; Dufour, S.; Staes, A.; Schymkowitz, J.; Rousseau, F.; Gallardo, R.; Delforge, M.; Van Dorpe, J.; Devos, S.; Impens, F.; Dendooven, A.
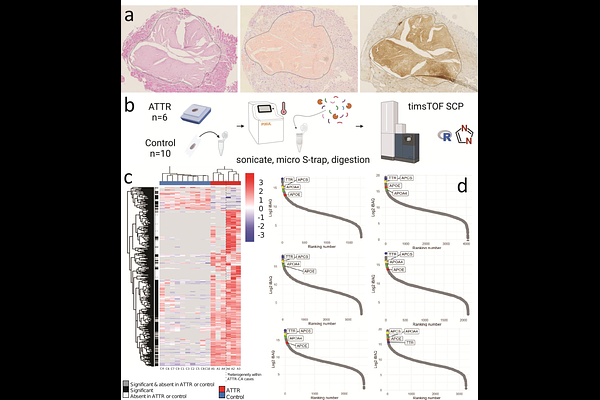
AbstractCardiac transthyretin amyloidosis (ATTR-CA) is caused by myocardial deposition of misfolded transthyretin, leading to progressive heart failure. Disease pathology, however, extends beyond passive amyloid deposition and also involves active processes such as extracellular matrix (ECM) remodeling and immune activation. Mass spectrometry (MS) is the gold standard for amyloid typing in diagnostics. Here, we applied quantitative MS-driven proteomics on formalin-fixed paraffin-embedded whole cardiac tissue sections from six ATTR-CA cases, ten unaffected controls and four AL-CA controls to investigate protein expression changes. In addition to transthyretin, over 500 proteins were upregulated in ATTR-CA biopsies, including complement and coagulation factors as well as extracellular matrix (ECM) remodeling proteins. Among these, members of the A Disintegrin and Metalloproteinase with Thrombospondin Motifs (ADAMTS) family, metalloproteinases (MMPs), and Tissue Inhibitor of Metalloproteinases (TIMP3) showed significant upregulation. These proteins are key regulators of ECM turnover and structural integrity. Immunohistochemistry confirmed ADAMTS4 enrichment in amyloid deposits, while TIMP3 showed strong expression in cardiomyocytes and weaker staining within amyloid deposits. Together, these findings indicate that ECM remodeling, alongside complement and coagulation activation, represents a reproducible feature of cardiac ATTR amyloidosis. Whole-tissue proteomics provides biological insights that extend beyond amyloid typing, with potential implications for biomarker discovery and therapeutic targeting in ATTR-CA.