FABP4 Couples Lipid Metabolism to PD-L1 Stabilization in Immunosuppressive Macrophages
FABP4 Couples Lipid Metabolism to PD-L1 Stabilization in Immunosuppressive Macrophages
Yu, J.; Shilyansky, J.; Avellino, A.; Hao, J.; Sun, Y.; Jiang, X.; Wang, Z.; Han, X.; Curry, M. A.; Sugg, S.; Li, B.
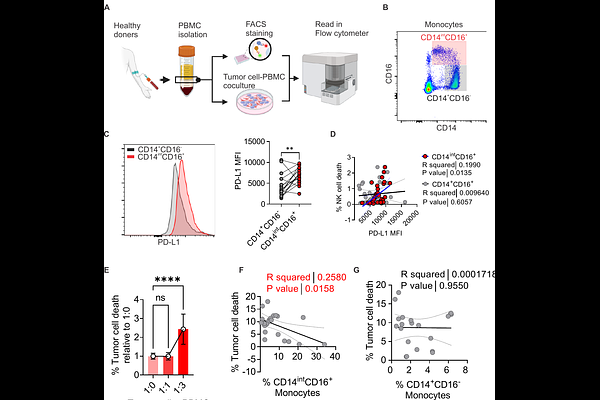
AbstractMetabolic dysregulation in obesity reshapes immune function, but how lipid signals drive immune suppression remains unclear. Here, we identify a FABP4/PD-L1 axis that links lipid metabolism to immune checkpoint regulation in monocytes and macrophages. Single-cell transcriptomics revealed a distinct FABP4high immunosuppressive macrophage subset enriched under high-fat diet (HFD) conditions, characterized by impaired antigen presentation and elevated PD-L1 expression. Mechanistically, palmitic acid (PA) induces FABP4 and promotes PD-L1 palmitoylation, leading to its stabilization on the cell surface independent of transcriptional regulation. FABP4 is essential for this process, which enables PD-L1 surface stabilization, immunosuppression and mammary tumor progression. In humans, a conserved CD14intCD16+; monocyte population exhibits elevated FABP4/PD-L1 signaling and correlates with obesity and invasive breast cancer. These findings establish PD-L1 as a metabolically regulated protein and reveal a mechanism by which lipid excess drives immune evasion, suggesting that targeting FABP4 may enhance responses to immune checkpoint blockade.