Conformation-Dependent Donor Selectivity in the Xanthan Gum Glycosyltransferase GumK Revealed by AI-Based Docking
Conformation-Dependent Donor Selectivity in the Xanthan Gum Glycosyltransferase GumK Revealed by AI-Based Docking
Luciano, D.; Alenfalk, T.; Courtade, G.
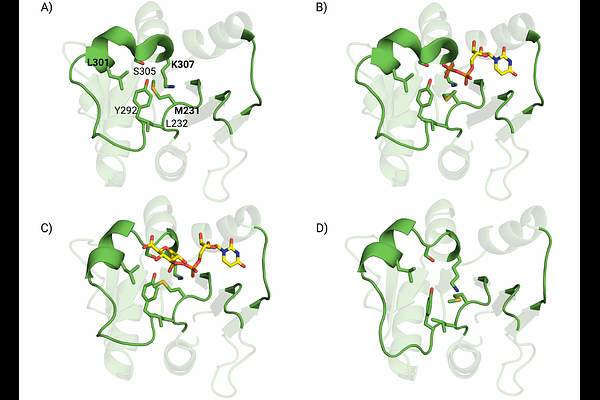
AbstractThe interdomain flexibility of GT-B fold glycosyltransferases regulates substrate binding and catalysis, yet the role of local structural variations in donor substrate specificity remains unclear. GumK, a GT70 enzyme from Xanthomonas campestris, exhibits local plasticity within its donor-binding pocket. We classify this plasticity into two conformational states, defined by the presence (closed state) or absence (open state) of a conserved hydrophobic interaction stabilizing the pocket. Using the AI-enhanced docking approach GNINA, we investigated the relationship between these states and substrate specificity by comparing UDP-glucuronate with various acidic and neutral substrate analogs. While docking scores showed limited discrimination among substrates, distance-based analysis between the sugar C6 atom and Lys307 revealed conformation-dependent trends. In the open state, negatively charged sugars preferentially interact with Lys307 via their carboxylate groups. Conversely, the closed state favors interactions with the pyrophosphate moiety. These results are consistent with physics-based simulations and suggest that donor specificity arises from the interplay between substrate chemistry and binding-site plasticity rather than from a single rigid binding mode. This study demonstrates how AI-driven docking, combined with an explicit representation of conformational states, can provide mechanistic insights into flexible enzymes and provide a rapid strategy for screening potential mutants.