Enhancing the detection of HTT1a with neoepitope antibodies in mouse models of Huntington's disease
Enhancing the detection of HTT1a with neoepitope antibodies in mouse models of Huntington's disease
Osborne, G. F.; Smith, E. J.; Sathasivam, K.; Kang, Z.; Nita, I. M.; Canibano-Pico, M.; Phillips, J.; Bates, G.; Landles, C.
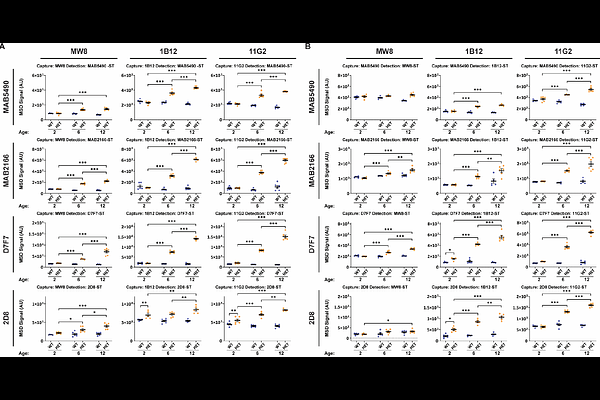
AbstractHuntington's disease is an inherited neurodegenerative disorder caused by a CAG repeat expansion in exon 1 of the Huntingtin (HTT) gene, encoding an expanded polyglutamine tract in the huntingtin (HTT) protein. The pathogenic CAG repeat of HTT is unstable and undergoes progressive somatic expansion in specific brain cells and peripheral tissues throughout life. Genes involved in DNA mismatch repair pathways, which promote repeat expansion, have been identified as genetic modifiers of the disease. Consequently, the rate of CAG repeat expansion is a key determinant driving the age of onset and disease progression. As the CAG repeat expands, alternative processing of HTT pre-mRNA increasingly favours production of the HTT1a transcript, which encodes the highly pathogenic and aggregation-prone HTT1a protein. This process provides a mechanistic link between CAG repeat expansion and disease pathogenesis, as increased HTT1a production accelerates HTT aggregation and neuronal dysfunction. HTT1a has previously been detected in Huntington's disease mouse models by using immunoprecipitation coupled with western blotting, homogeneous time-resolved fluorescence (HTRF) and Meso Scale Discovery (MSD) bioassays, and immunohistochemistry. These approaches were developed using MW8, a neoepitope antibody that specifically recognizes the C-terminus of HTT1a. MW8 is a relatively weak antibody with limited detection sensitivity. To generate more robust HTT1a-specific reagents, two novel recombinant antibodies, 1B12 and 11G2, have been developed for evaluation. Using an allelic series of knock-in (HdhQ20, HdhQ50, HdhQ80, HdhQ111, CAG140 and zQ175) mice, alongside transgenic YAC128 and N171-82Q models, we extensively evaluated and compared the performance of MW8, 1B12 and 11G2. We demonstrate that 1B12 and 11G2 function as HTT1a-specific neoepitope antibodies by immunoprecipitation with western blotting, and by immunohistochemistry. To enhance HTT1a detection using HTRF and MSD technology platforms, we further evaluated the performance of 1B12 and 11G2 in HTT bioassays using cortical lysates from zQ175 and YAC128 mice. In zQ175 mice, enhanced detection of aggregated HTT1a by HTRF and MSD revealed that HTT fragments longer than HTT1a can be incorporated into HTT1a-containing aggregates. The most sensitive assays were subsequently applied across the allelic series of knock-in mice to assess the effect of polyglutamine length on bioassay performance. For optimal sensitivity, we recommend the preferential use of 1B12 for HTRF assays and 11G2 for MSD assays. Collectively, these findings establish 1B12 and 11G2 as robust antibodies to reliably detect and track HTT1a pathology in vivo and promotes the replacement of previously used MW8-based experimental approaches.