Organotypic artery-graft culture enables label-free multiphoton tracking of remodeling that links to long-term graft microarchitecture
Organotypic artery-graft culture enables label-free multiphoton tracking of remodeling that links to long-term graft microarchitecture
Maestas, D. R.; Murphy, T.; Martinet, K. M.; Moyston, T.; Min, L. X.; Behrangzade, A.; Pemberton, B. J.; Ye, S.-H.; Hussey, G. S.; Azhar, M.; Wagner, W. R.; Vande Geest, J. P.
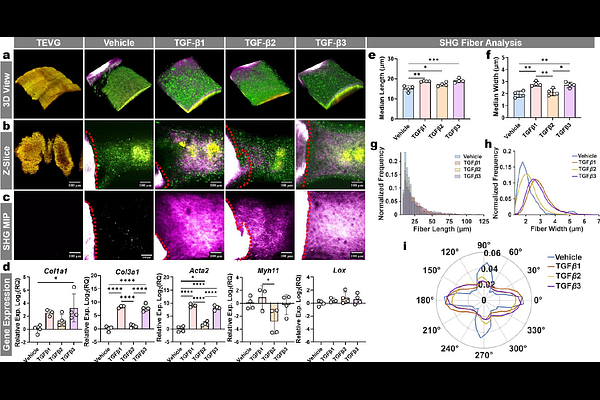
AbstractTissue-engineered vascular grafts (TEVGs) are promising alternatives for blood vessel replacement, yet their clinical translation is constrained by resource-intensive implantation studies. While necessary, implantation models obscure processes that govern long-term outcomes, such as time-resolved microscale remodeling. Here, we develop an organotypic culture that couples rat aortic explants with acellular grafts in a cylindrical geometry. Harnessing label-free non-destructive multiphoton imaging, we longitudinally assess fibrillar collagen via second harmonic generation (SHG) and simultaneously detect cellular and TEVG states by their respective two-photon excited fluorescence (2PEF). Fiber quantification unveils an evolving collagen architecture that is verified by orthogonal staining. We verify compatibility with common biomaterials, murine fluorescent reporters, and terminal assays. We demonstrate the platform perturbation sensitivity via treatment with transforming growth factor-{beta} isoforms (TGF-{beta}1/-{beta}2/-{beta}3) and identify shifts in fiber distributions coinciding with differential gene expression. The architectural fiber distributions observed in culture show agreement with 6-month interpositional aortic explants across two graft designs, supporting a trajectory-matching readout of remodeling. Collectively, this platform provides an accessible decision-informing tool to assess biomaterial scaffold designs prior to surgical implantation.