Comprehensive study on ferredoxin isoforms in the cyanobacterium Synechocystis sp. PCC 6803
Comprehensive study on ferredoxin isoforms in the cyanobacterium Synechocystis sp. PCC 6803
Boehm, M.; Svedruzic, D.; Lubner, C. L.; Appel, J.; Mulder, D. W.; Kisgeropoulos, E.; Hueren, V.; Spengler, K.; Bharadwaj, V.; Guo, Z.; Ledinina, A. E.; Deobald, D.; Adrian, L.; King, P. W.; Gutekunst, K.
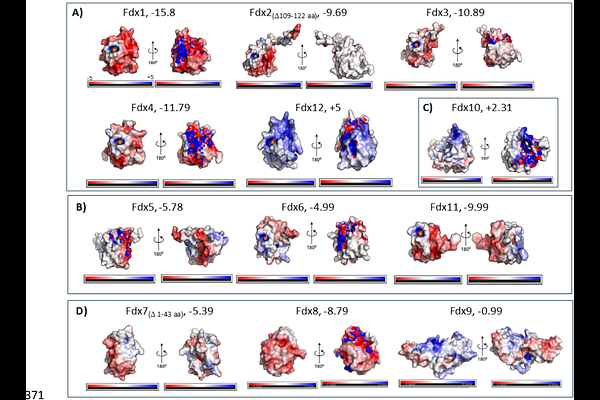
AbstractFerredoxins are central to cellular metabolism by mediating electron flow in energy conversion reactions. The focus of this study was to systematically examine twelve ferredoxin and ferredoxin-like proteins from Synechocystis sp. PCC 6803 to identify their properties, activities, and functions in electron transfer. Using electron paramagnetic resonance spectroscopy, we detected cluster types consistent with major ferredoxin families including plant-type [2Fe2S], adrenodoxin, thioredoxin, and bacterial-type [4Fe4S] ferredoxins. In addition, we found that the ssr3184 ferredoxin-like protein exchanged between a [3Fe4S] or a [4Fe4S] cluster, pointing to a possible functional change in response to changes in oxygen or cellular redox poise. Electrochemical measurements demonstrated that these ferredoxins constitute a broad potential window, from -243 mV to - 520 mV vs SHE. Investigations on their capacity to support electron-transfer focused on reactions with two major redox hubs: Photosystem I and pyruvate:ferredoxin oxidoreductase and included testing of binding interactions with nitrite reductase. Expression profiling under multiple environmental conditions was also used to predict function and revealed distinct regulatory patterns. Collectively, these findings identified a group of core ferredoxins that directly support photosynthetic electron transfer, and more specialized ones that may serve other functions. In summary, Synechocystis utilizes a suite of ferredoxins to maintain cellular redox homeostasis under dynamic environmental conditions.