Mast cell desensitization induces a distinct IgE-dependent transcriptional program associated with immune regulation
Mast cell desensitization induces a distinct IgE-dependent transcriptional program associated with immune regulation
Lopez-Sanz, C.; Nunez-Borque, E.; Ruiz-Sanchez, A.; Sevilla-Montero, J.; Garcia-Civico, A. J.; Alvarez-Garrote, S.; Zamora-Dorta, M.; Sanchez-Martinez, E.; Moreno-Serna, L.; Mamani-Huanca, M.; Villasenor, A.; Barber, D.; Balsa, E.; Munoz-Calleja, C.; Ojeda, P.; Blanco, C.; Rodriguez del Rio, P.; Sanchez-Madrid, F.; Jimenez-Saiz, R.
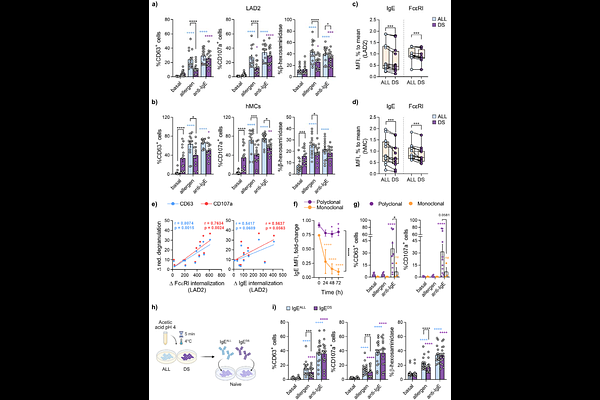
AbstractAllergen-driven IgE-mast cell (MC) activation is a central feature of allergic diseases, whose prevalence continues to increase worldwide. Allergen immunotherapy (AIT) is currently the only disease-modifying treatment and induces a state of MC hyporesponsiveness termed desensitization; however, its underlying molecular mechanisms remain incompletely understood and whether this state reflects passive signal attenuation or active cellular reprogramming remains unresolved. Here, we define the molecular landscape of MC desensitization using a human polyclonal platform that captures the physiological diversity of allergen-specific IgE. Desensitization reduced degranulation in an allergen-specific manner and induced progressive internalization of allergen-specific IgE. Although early steps were associated with LAT phosphorylation, subsequent allergen challenge failed to propagate activation to distal IgE/Fc{varepsilon}RI effectors, revealing selective signaling uncoupling. Transcriptomic profiling uncovered a distinct transcriptional program comprising 168 upregulated genes enriched in immunoregulatory pathways and largely non-overlapping with classical activation signatures. This reprogramming occurred despite minimal alterations in mitochondrial respiration and selective impairment of allergen-induced glycolysis. Functionally, desensitized MCs enhanced allergen-driven proliferation of memory CD4 T cells. Together, these findings demonstrate that MC desensitization is not merely passive hyporesponsiveness but involves time-dependent allergen-specific IgE internalization, selective signal propagation, and a unique immunoregulatory transcriptional imprint that may contribute to tolerance during AIT.