Natural variation in transplacental transfer efficiency exposes distinct transcriptional network architectures of PFAS effects on birth weight and gestational age
Natural variation in transplacental transfer efficiency exposes distinct transcriptional network architectures of PFAS effects on birth weight and gestational age
Bresnahan, S. T.; Yong, H. E. J.; Drelichman, M. G.; Campbell, S. N.; Trapse, A. E.; Romo, G. R.; Cellini, C. M.; Lopez, S.; Chan, J. K.; Chan, S.-Y.; Elkin, E. R.; Bhattacharya, A.; Huang, J. Y.
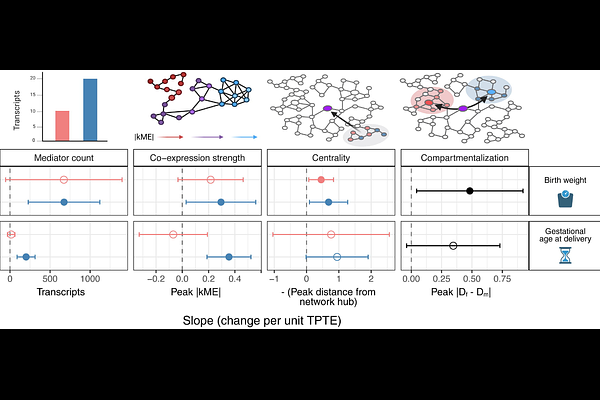
AbstractPer- and polyfluoroalkyl substances (PFAS) are environmental contaminants that produce heterogeneous effects on perinatal outcomes despite chemical similarity. Natural variation in transplacental transfer efficiency (TPTE, the degree to which compounds cross the placental barrier) presents a mechanistic lens for understanding this heterogeneity, but whether TPTE systematically shapes the transcriptional pathways linking exposure to outcomes has not been tested. Using isoform-resolved placental transcriptomics from n = 124 term deliveries with eight PFAS measured in maternal and cord blood, validated against patient-derived placental explants (n = 18) using a tissue-specific long-read transcriptome assembly, we show that PFAS influence perinatal outcomes primarily through co-expression network hubs rather than differentially expressed features, indicating that network position, not fold-change magnitude, determines mediator importance. Leveraging TPTE as a natural experiment, we find that direct fetal exposure recruits increasingly numerous and tightly coordinated transcriptional mediators for both birth weight and gestational age, but the two outcomes diverge in network architecture: network centrality and maternal-fetal compartmentalization scale with TPTE exclusively for birth weight, while gestational age shows no such topological reorganization. These outcome-specific patterns are detectable only at the transcript level, as gene-level aggregation masks systematic TPTE-network relationships. This framework distinguishes which perinatal outcomes are mechanistically vulnerable to fetal exposure dose, with implications for risk assessment and therapeutic target identification for environmental contaminants.