Resolution of the D4Z4 repeat responsible for facioscapulohumeral muscular dystrophy with HiFi sequencing
Resolution of the D4Z4 repeat responsible for facioscapulohumeral muscular dystrophy with HiFi sequencing
Chen, X.; Lemmers, R. J. L. F.; Kronenberg, Z.; Devaney, J. M.; Noya, J.; Berlyoung, A. S.; Yusuff, S.; Lynch, S.; Nykamp, K.; Lyndy, A. S.; Dolzhenko, E.; van der Maarel, S. M.; Eberle, M. A.
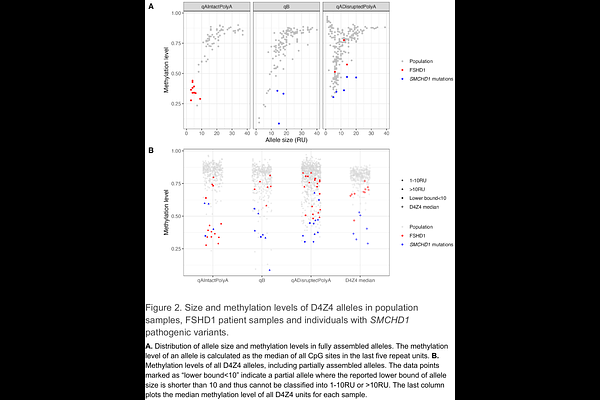
AbstractThe D4Z4 macrosatellite repeat encompasses some of the most difficult-to-resolve disease-related variations in the human genome. D4Z4 has a repeat unit of 3.3 kb (encoding the DUX4 gene) that is present in up to 100 copies on two chromosomes (4 and 10), while DUX4 can only be expressed in somatic cells from the permissive A haplotype that usually occurs on chromosome 4. Facioscapulohumeral muscular dystrophy (FSHD) is caused by chromatin relaxation and ectopic expression of DUX4 in skeletal muscle, mediated by contraction of D4Z4 to 1-10 copies (FSHD1, 95% of FSHD cases) or mutations in chromatin factor genes such as SMCHD1 (FSHD2, 5% of FSHD cases). Due to its large size, disease specific haplotypes and sequence homology between chromosomes, D4Z4 is challenging to resolve by current sequencing technologies. We report a computational tool, Kivvi, to genotype D4Z4 using PacBio whole-genome long-read sequence data. Kivvi detects all D4Z4 alleles in a sample, reporting the repeat size, chromosome (4 vs. 10), distal haplotype (A vs. non-permissive haplotypes) and the methylation level of each allele. We validated Kivvi against gold standard assays for FSHD diagnostics, detecting 100% of contracted alleles and correctly classifying 90% of noncontracted alleles. We showed differential methylation signals between FSHD1 and candidate FSHD2 samples. We profiled D4Z4 across 601 individuals from five ancestral populations, revealing extensive genetic diversity. We identified common haplotypes of D4Z4 alleles and characterized hybrid repeat units, hybrid repeat arrays, and translocation alleles. Combined with HiFi long reads, Kivvi enables the consolidation of multiple FSHD assays into a single workflow and facilitates the discovery of novel genetic modifiers of FSHD through population-scale studies.