Disease context dictates the cellular targets of IL-17 in inflammatory skin disease
Disease context dictates the cellular targets of IL-17 in inflammatory skin disease
Cavagnero, K. J.; Jo, H.; Li, F.; Aguilera, C.; Fox, J.; Kirma, J.; Bogel, R.; Kahlenberg, J. M.; Tsoi, L. C.; Gudjonsson, J. E.; Gallo, R. L.
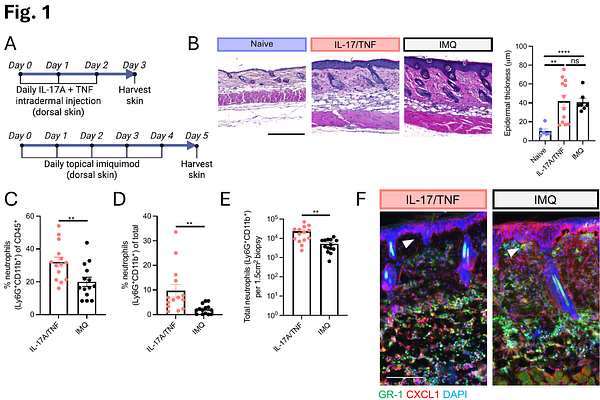
AbstractTherapeutic blockade of IL-17 and TNF can effectively treat inflammatory skin diseases such as hidradenitis suppurativa and psoriasis, yet the relative importance of the different cell types that respond to IL-17 and TNF remains unresolved. Keratinocytes are viewed as the dominant effector cells, whereas fibroblasts have recently emerged as important contributors. In mice, topical imiquimod induces IL-17- and TNF-dependent skin inflammation and is frequently used to model psoriasis. Here, we demonstrate that intradermal injection of recombinant IL-17 and TNF elicits skin inflammation with features of hidradenitis suppurativa, including a gene expression program that is distinct from psoriasis and imiquimod-induced inflammation. Single-cell transcriptomic network analysis identified dermal fibroblasts as the dominant cell communication hub in hidradenitis suppurativa and in mice injected with IL-17 and TNF. In contrast, fibroblasts and keratinocytes both show strong network involvement in psoriasis and in mice challenged with imiquimod. Cell-type-specific deletion of IL-17 receptor A in mice revealed that imiquimod-induced inflammation depends equally on IL-17 signaling in fibroblasts and keratinocytes, whereas inflammation induced by intradermal IL-17 and TNF only requires fibroblasts to recognize IL-17 and is independent of keratinocyte IL-17 sensing. Single-cell transcriptomic analysis of these conditional knockout mice further demonstrated that keratinocytes and fibroblasts activate divergent and disease-dependent transcriptional programs following activation by IL-17. Together, these findings introduce a new conceptual framework wherein IL-17 signaling is routed through distinct cellular and molecular pathways depending on disease context and establish complementary experimental systems for interrogating type 17 skin inflammation.