Structural basis of substrate recognition for proteasome degradation by prokaryotic ubiquitin-like protein ligase PafA
Structural basis of substrate recognition for proteasome degradation by prokaryotic ubiquitin-like protein ligase PafA
Plourde, A.; Uday, A. B.; Forrester, T. J. B.; Zeytuni, N.; Vahidi, S.
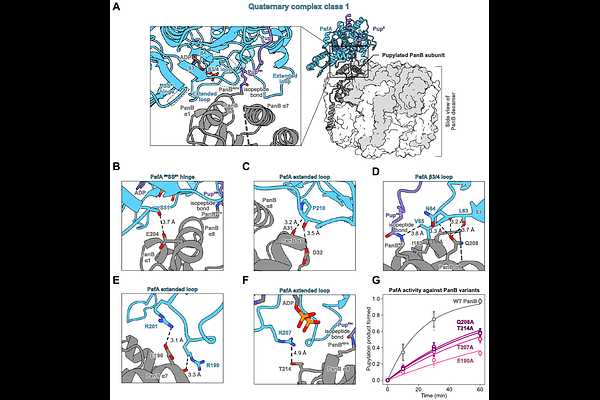
AbstractSelective protein degradation in some bacteria is performed by the Pup-proteasome system, in which the ligase PafA tags hundreds of substrates for proteasomal degradation. How a single enzyme achieves such broad substrate specificity in the absence of conserved sequence motifs has remained unclear. Here we determine structures of PafA in complex with a pupylated substrate and show that substrate recognition is mediated by a minimal and highly distributed interface. PafA samples an ensemble of closely related conformations that collectively position the target lysine residue for modification. This recognition mechanism arises from a combination of structured contacts and dynamic elements on both the enzyme and substrate, enabling geometric compatibility rather than sequence-specific interactions. These findings reveal an ensemble-driven mechanism of molecular recognition that explains how broad substrate specificity is achieved and provides a framework for understanding selective protein degradation in prokaryotes.