Homologues of the inner-membrane LPS transport proteins are required for sphingolipid transport in Caulobacter crescentus
Homologues of the inner-membrane LPS transport proteins are required for sphingolipid transport in Caulobacter crescentus
Uchendu, C. G.; Isom, G. L.; Klein, E. A.
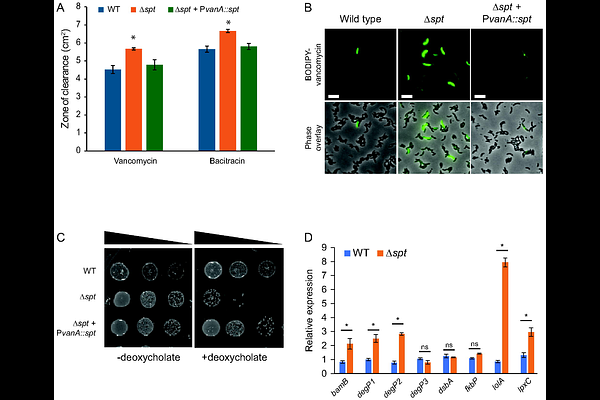
AbstractRecent elucidation of the bacterial sphingolipid synthesis pathway has revealed that these lipids are produced by a range of taxonomically diverse species. In contrast to the biosynthetic pathways, the mechanism by which sphingolipids are transported from the inner membrane to the cell surface in Gram-negative bacteria remains a mystery. Here, we identify and characterize paralogs of the well-characterized lipopolysaccharide (LPS) inner membrane ABC transporter proteins encoded within the sphingolipid locus. Using Caulobacter crescentus as a model system, we analyzed three putative inner membrane proteins with homology to LptF, LptG, and LptC. Deletion of these genes was lethal, likely due to the accumulation of anionic sphingolipids in the inner membrane. We further show that the LptF and LptG homologues form a complex like their LPS counterparts and discover that they interact with the LPS ATPase LptB. Together, our data suggest that ceramide transport to the outer membrane is facilitated by an ABC transporter consisting of a sphingolipid-specific LptFG homolog coupled to the LPS LptB, supporting a model in which sphingolipid transport partially converges with the LPS transport system. Together, these findings reveal an unexpected evolutionary relationship between sphingolipid and lipopolysaccharide transport.