ENPP1 buffers extracellular cGAMP in brown adipose tissue to limit insulin resistance
ENPP1 buffers extracellular cGAMP in brown adipose tissue to limit insulin resistance
Wang, S.; Guo, Y.; An, W.; Lee, M.; Li, Y.; Sudaryo, V.; Grenot, G.; Skariah, G.; Reghupaty, S. C.; Young, S.; Bai, X.; Svensson, K. J.; Li, L.
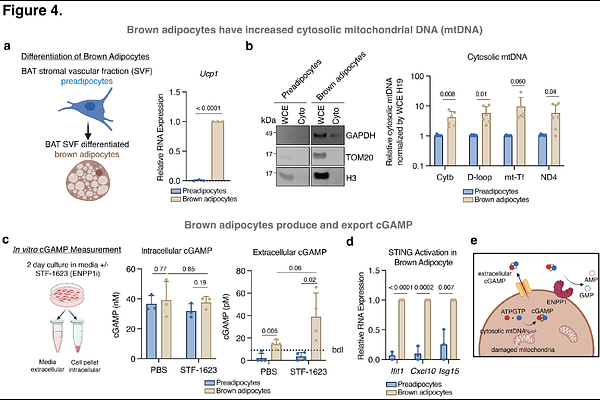
AbstractThe ectonucleotide pyrophosphatase/phosphodiesterase 1 (ENPP1) has long been linked with metabolic diseases, with the common ENPP1 K173Q (historically K121Q) variant conferring increased risk for type 2 diabetes (T2D). However, the mechanistic basis of this association has remained unclear. Here, we demonstrate that the K173Q variant has decreased cGAMP hydrolysis activity, suggesting that this loss of enzymatic function could contribute to its pathogenesis. Using a cGAMP-hydrolysis-deficient knock-in mouse (Enpp1H362A), we show that selective loss of this activity leads to a primary defect in energy expenditure and exacerbates high-fat diet (HFD)-induced weight gain and insulin resistance. An unbiased in vivo glucose-uptake screen reveals brown adipose tissue (BAT) as a focal site of metabolic impairment, characterized by profound extracellular cGAMP accumulation and a selective failure of insulin-stimulated glucose uptake. Mechanistically, we demonstrate that nutrient excess drives mitochondrial DNA leakage in brown adipocytes, triggering cGAMP production and export. Excess cGAMP directly propagates STING-dependent suppression of glucose uptake and lipogenesis in brown adipocytes. Additionally, when ENPP1-mediated clearance is compromised, extracellular cGAMP acts as a paracrine immunotransmitter that remodels the BAT microenvironment by recruiting and polarizing macrophages toward an M1-like phenotype. Together, our findings nominate the impaired ENPP1-dependent buffering of extracellular cGAMP as one mechanism by which ENPP1 variants influence metabolic homeostasis.