Efficient coding explains altered neural representations elicited by subtle sensory lesions
Efficient coding explains altered neural representations elicited by subtle sensory lesions
M. Fuentes, J. A.; Undurraga, J.; Schaette, R.; McAlpine, D.
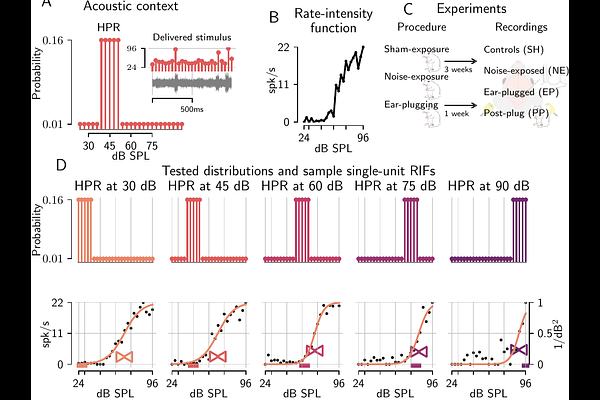
AbstractSensory systems must represent a vast range of stimulus dimensions and energy whilst subject to metabolic constraints. Efficient-coding theory predicts that neural adaptation re-allocates a relatively limited range of neural activity toward the most informative stimulus values, but it is unclear how subtle peripheral lesions shift this operating point in central circuits. Hearing is a stringent test because sound level varies enormously across environments, yet clinical assessment still relies heavily on tone-detection thresholds that can miss listening deficits in noise. We analyzed extracellular recordings from single neurons in the gerbil auditory midbrain across 14 animals in four experimental groups exposed to unfolding distributions of sound intensities drawn either uniformly from a wide range (24--96 decibels) of sound pressure levels or from contexts in which 80% of levels were restricted to a 12-decibel high-probability range. For each context we summarized each neuron's rate--intensity input--output function by an effective threshold and gain, and we interpreted the resulting threshold--gain distributions with an information--cost model that trades bits of stimulus information against a penalty on mean spiking. Noise exposure consistent with loss of synapses between inner-ear cells and auditory nerve fibers altered gain modulation across acoustic contexts, with noise-exposed animals showing compressed gain adjustments relative to controls; within the information--cost framework, the clearest hidden-hearing-loss effect was a quiet-context utility advantage concentrated in the low-threshold neural population, whereas moderate-to-loud contexts showed weaker or absent group differences. Temporary conductive attenuation caused by ear-canal plugging shifted effective thresholds to higher sound levels, with incomplete recovery after plug removal; the corresponding optimization-prior trajectories were consistent with incomplete rapid renormalization but were weaker than the hidden-hearing-loss effect. These results support an efficient-coding interpretation of altered central auditory representations after subtle lesions and provide a quantitative, context-based framework for comparing mechanisms of hearing difficulty beyond threshold-only tests and Fisher information alone.