Development of semisynthetic blasticidin S analogs with potent and fast-killing anti-malarial activity
Development of semisynthetic blasticidin S analogs with potent and fast-killing anti-malarial activity
Fike, K. R.; Gannett, C.; Kiselka, A. M.; Tiller, K.; Ajasa, T.; Weger-Lucarelli, J.; Brown, A. M.; Lowell, A. N.; Klemba, M.
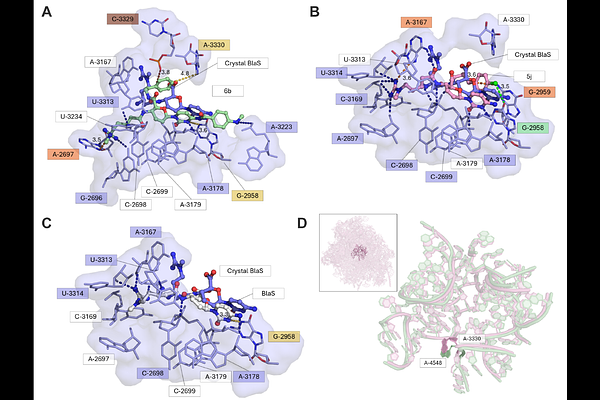
AbstractProtein synthesis represents an attractive target space for the development of anti-malarials with novel modes of action. Natural-product inhibitors of the eukaryotic 80S ribosome can have potent anti-malarial activity but are often poorly selective due to mammalian cytotoxicity. Blasticidin S (BlaS) is a microbially-produced natural product that broadly inhibits prokaryotic and eukaryotic protein synthesis by binding to the ribosomal peptidyl transferase center. In this study, we explored the potential for improving the anti-malarial potency and selectivity of the blasticidin S scaffold with semi-synthetic analogs that are modified at the C6' and C4 sites. The two best analogs were two orders of magnitude more potent than BlaS against Plasmodium falciparum drug-sensitive and -resistant lines while displaying low cytotoxicity towards mammalian cells. These analogs exhibited improved kinetics of inhibition of protein synthesis in cultured parasites and blocked the development of asexual stages expressing the plasmodial surface anion channel, a transporter required for nutrient acquisition and BlaS uptake. They also exhibited a dramatically improved speed of killing over BlaS. Molecular docking analysis revealed that these analogs are able to form more interactions with the P. falciparum ribosomal peptidyl transferase center than is BlaS, which is consistent with their increased potency. Together, these studies demonstrate the feasibility of generating BlaS analogs with potent anti-malarial activity and provide a roadmap for further development.