Phloem evolved gradually and asynchronously to xylem in early vascular plants
Phloem evolved gradually and asynchronously to xylem in early vascular plants
Cooper, L. M.; Hetherington, A. J.
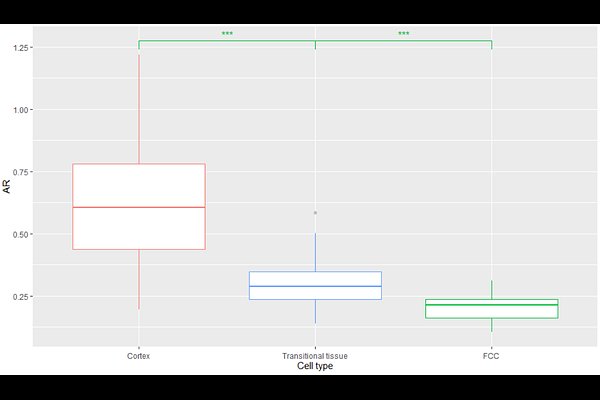
AbstractThe evolution of the water-conducting xylem and sugar-conducting phloem tissues were key innovations in land plant evolution, enabling the origin of long-distance transport networks (1). In extant vascular plants, phloem and xylem are linked functionally and always occur together (2), though their evolutionary origin is unclear. This uncertainty is owed to the greater fossilisation potential of lignified xylem tracheids compared to thin-walled phloem cells (3, 4, 5). Therefore, the fossil record of xylem is far more extensive than that of phloem, with the first definitive record of xylem being around 40 million years earlier (6) than phloem (7). This bias in the fossil record obscures characterisation of the origins of plant vasculature. In this study, this limitation is overcome by re-describing the 'phloem-like' tissues of exceptionally preserved plants from the 407-million-year-old Rhynie chert8-15. We report that this tissue differs markedly from the phloem of extant plants, and propose its identification as a tissue of food-conducting cells (FCCs). Major histological differences were observed in the fossil plants, including no evidence for a pericycle, which in extant species delimits vascular from ground tissues, and the FCCs of the Rhynie chert plants were significantly larger in diameter than phloem cells. These differences suggest that early vascular plants lacked true phloem. However, putative sieve pores in the FCCs of Asteroxylon mackiei were identified. This represents to our knowledge the earliest record of sieve pores in the fossil record. Our results suggest an evolutionary scenario in which phloem features assembled gradually within FCCs, asynchronous to the evolution of xylem.