Exploiting HLA-II Promiscuity via Peptide Terminal Overhang Recognition for Pan-Allelic and Tumor-Selective AML Immunotherapy
Exploiting HLA-II Promiscuity via Peptide Terminal Overhang Recognition for Pan-Allelic and Tumor-Selective AML Immunotherapy
Fukao, S.; Zheng, E.; Ihara, F.; Matsunaga, Y.; Ohashi, Y.; Han, D.-H.; Wei, X.; Hasegawa, K.; Burt, B. D.; Saso, K.; Ly, D.; Butler, M.; Minden, M.; Kagoya, Y.; Hirano, N.
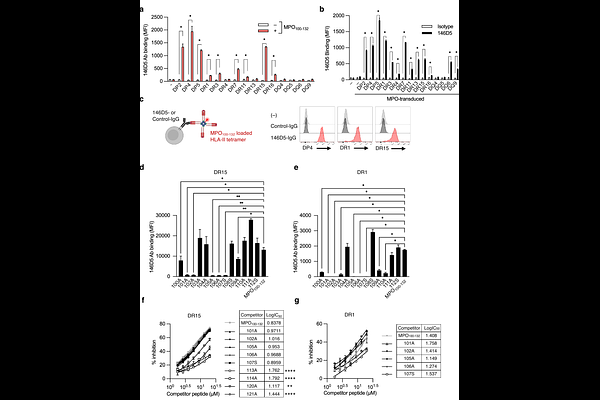
AbstractAntibodies targeting peptides presented by human leukocyte antigen (HLA) molecules expand the therapeutic landscape by enabling recognition of intracellular antigens. While most efforts have focused on allele-restricted peptides presented by HLA class I (HLA-I), HLA class II (HLA-II) epitopes remain underexplored despite their potential for promiscuous presentation. Acute myeloid leukemia (AML) is characterized by high expression of both HLA-II and the myeloid lineage antigen myeloperoxidase (MPO). Here, we identified an MPO-derived epitope (MPO100-132) that is promiscuously presented by multiple HLA-II molecules. We generated a MPO100-132-specific antibody (146D5) that recognizes the N-terminal overhang of this peptide independent of specific HLA contacts, enabling pan-allelic recognition. Engineered into bispecific T cell engagers (BiTEs), this antibody mediated robust cytotoxicity against primary AML samples across diverse HLA-II backgrounds. Crucially, 146D5-based BiTEs selectively spared normal myeloid cells, indicating that the MPO100-132 peptide, derived from the MPO propeptide, was functionally undetectable in normal myeloid cells, providing a significant safety window. In vivo, the MPO-targeting BiTE demonstrated potent antitumor activity and prolonged survival in AML xenograft models. Our findings identify peptide terminal overhangs as an actionable class of antibody targets and introduce a strategy to exploit HLA-II promiscuity for broadly applicable HLA-dependent but allele- agnostic immunotherapies.