GEF me a break: the consequences of freezing Rho guanine-nucleotide exchange factor catalytic domains
GEF me a break: the consequences of freezing Rho guanine-nucleotide exchange factor catalytic domains
Anderson, L. K.; Barpal, E.; Mendoza, H.; Cash, J. N.
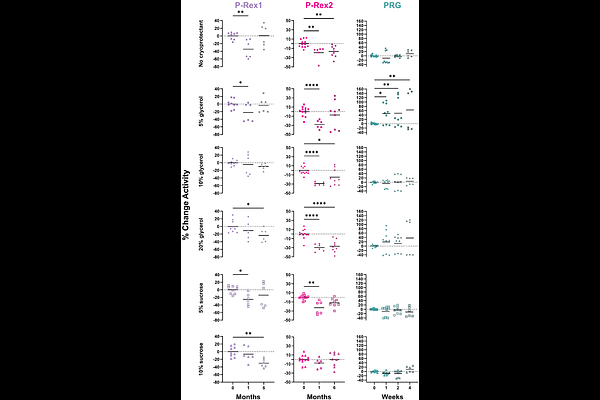
AbstractPurified proteins are routinely flash frozen for use in functional and structural studies, providing a convenient way to reproduce results across complex experiments. Rho guanine-nucleotide exchange factors (RhoGEFs) are no exception to this practice, yet the effects of freezing on their activity and stability remain largely uncharacterized. This gap potentially affects the characterization of these important enzymes and how results are interpreted with respect to their prospective use as therapeutic targets. Here, we tested the isolated DH/PH tandems of P-Rex1, P-Rex2, and PRG under different cryoprotectant conditions and monitored activity and thermostability over time after flash freezing. Our results show a clear divergence between the activity of fresh and frozen purified RhoGEF protein samples in as little as one week for some conditions. Specifically, the variability in data collected on frozen samples was greatly increased. Despite these differences, thermostability seems to be preserved for much longer timepoints across RhoGEFs. Moreover, despite eventual changes in both activity and thermostability with respect to freezing, there are no obvious changes in global conformation between fresh and frozen samples of the isolated P-Rex2 DH/PH tandem. From our data, there are few generalizable trends between the different RhoGEFs and no single cryoprotective agent tested was a silver bullet to preserve both activity and thermostability across RhoGEFs. Overall, our findings emphasize the unpredictable effects of freezing RhoGEFs. As such, RhoGEF freezing should be carefully characterized for each protein and critically viewed when comparing analyses between different studies.