Small Molecule Regulation of CLOCK:BMAL1 DNA Binding Activity
Small Molecule Regulation of CLOCK:BMAL1 DNA Binding Activity
Sharma, D.; Boral, S.; West, E.; Kressman, M.; Franco, I.; Amezcua, C. A.; Tripathi, S.; Lee, H.-W.; Favaro, D. C.; Gardner, K. H.; Partch, C. L.
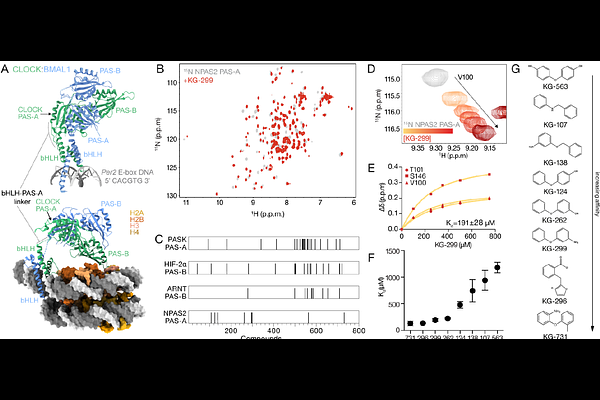
AbstractCLOCK:BMAL1 is a bHLH-PAS transcription factor complex that utilizes its bHLH (basic helix-loop-helix) domains to bind E-box motifs in DNA and tandem PAS (PER-ARNT-SIM) domains to heterodimerize and interact with regulatory proteins to generate circadian rhythms. PAS domains are evolutionarily conserved modules that frequently bind small molecule ligands within buried cavities to perform sensory and signal transduction functions. CLOCK and BMAL1 PAS domains have cavities that could be leveraged to regulate the transcription factor, and consequently, the circadian clock. Using NMR spectroscopy, we identified small molecules that bind within a cavity inside the PAS-A domain of CLOCK and its paralog NPAS2, which sits at an important flexible junction in the structured core of the heterodimer. We identified a gatekeeping mutant in the core of CLOCK PAS-A that significantly decreased ligand binding affinity. High-pressure NMR studies showed that ligand binding or the gatekeeping mutant significantly stabilized the domain. Finally, we showed that ligands induced dose-dependent displacement of CLOCK:BMAL1 from DNA in vitro. Together, these data demonstrate that small molecules can regulate DNA binding by the circadian transcription factor CLOCK:BMAL1 through occupancy of a PAS domain cavity.