Reduced LACTB expression in myeloid cells is associated with elevated succinylcarnitine levels and reduced Alzheimers disease risk.
Reduced LACTB expression in myeloid cells is associated with elevated succinylcarnitine levels and reduced Alzheimers disease risk.
Romero-Molina, C.; Gomez-Gutierrez, R.; See, W. Y.; Patel, T.; Davtyan, H.; Ma, J.; Xu, Q.; Sewell, M.; Allton, K.; McReynolds, M.; Calderon, O.; Lightfoot, Y. L.; Bommer, G.; Cruchaga, C.; Blurton-Jones, M.; Ray, W. J.; Marcora, E.; Goate, A. M.
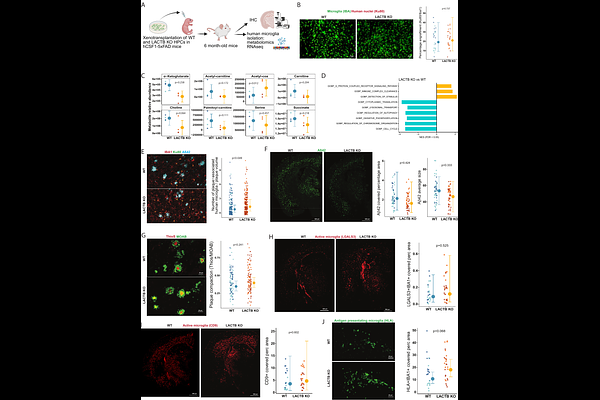
AbstractBackground: Lactamase B (LACTB) is a mitochondrial protease associated with cancer progression and lipid metabolism. LACTB is located in an AD locus and has been associated with Alzheimers Disease (AD) in a proteomic study. Methods: We performed Mendelian randomization (MR) to evaluate the relationship between LACTB expression, succinyl-carnitine, and AD risk. We generated LACTB knock-down (KD) THP1 macrophages, LACTB knock-out (KO) iPSC-derived microglia and LACTB enzymatically-dead (ED) mice. The impact of LACTB downregulation in myeloid cells was characterized via transcriptomics, metabolomics, lipidomics, and functional assays. Finally, human LACTB KO microglia precursors were xenotransplanted into the brains of amyloid-pathology mice to assess in vivo interactions with amyloid plaques. Results: MR analyses revealed that lower LACTB expression in myeloid cells reduces AD risk and is genetically associated with increased levels of succinylcarnitine, a metabolite that independently correlates with reduced AD risk. We identified LACTB as a primary enzyme responsible for succinylcarnitine hydrolysis. Transcriptional and functional studies showed that loss of LACTB enhances OXPHOS, reduces protein synthesis, and alters lipid profiles. LACTB expression was upregulated following IFN/TNF stimulation, and its loss modified efferocytosis-related functions under inflammatory conditions. In vivo, xenotransplanted human LACTB-KO microglia exhibited enhanced association with amyloid plaques compared to controls. Conclusions: Our findings define a previously unrecognized axis linking LACTB and succinylcarnitine to myeloid cell function and AD susceptibility. Given the druggability of LACTB and the potential for succinylcarnitine to serve as a translational biomarker, this pathway represents a promising therapeutic target for modulating neuroinflammation in AD.