Crystal structure and molecular dynamics simulations of rademikibart Fab-IL-4Rα complex reveal biochemical basis for next-generation potent IL-4Rα inhibition in type 2 allergic and inflammatory diseases
Crystal structure and molecular dynamics simulations of rademikibart Fab-IL-4Rα complex reveal biochemical basis for next-generation potent IL-4Rα inhibition in type 2 allergic and inflammatory diseases
Shi, Y.; Nolden, K.; Ho, M.; Li, H.; Batista, V. S.; Collazo, R.; Bunick, C. G.
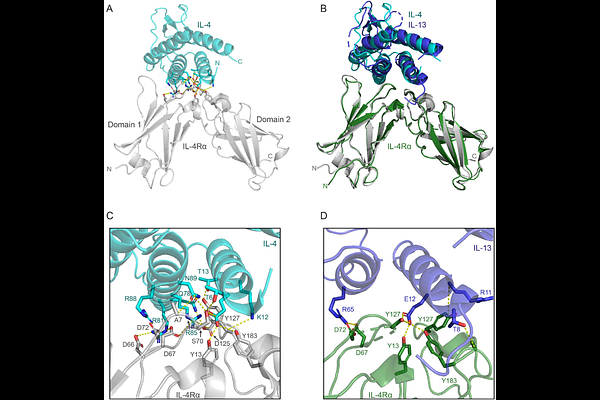
AbstractRademikibart (CBP-201) is a human monoclonal antibody with higher binding affinity to IL-4R compared to dupilumab. Dupilumab is a first-generation interleukin-4 receptor alpha (IL-4R) inhibitor for treating IL-4R-dependent inflammatory disorders, including several dermatologic and respiratory conditions. Rademikibart, however, demonstrated better inhibition of STAT6 intracellular signaling in vitro and similar potency in inhibiting both IL-4 induced TARC release and IL-4 induced B cell activation. To further characterize the molecular function of rademikibart and its differentiation from dupilumab, we determined the crystal structure of the rademikibart fragment antigen binding (Fab) bound to IL-4R at 2.71 [A] resolution and compared this to the 2.82 [A] resolution structure of dupilumab Fab bound to IL-4R. The rotation angle between dupilumab and rademikibart bound to IL-4R is 54.88{degrees}. This rotation enables the binding epitopes of rademikibart, but not dupilumab, on IL-4 to overlap more closely with the conserved binding interface naturally utilized by IL-4 and IL-13 cytokines. Molecular dynamics (MD) studies on rademikibart and dupilumab bound to IL-4R examined the stability of the complexes and effects of amino acid mutations on receptor complex formation. MD simulations demonstrated that the third interface loop (residues 145 to 153 in domain 2) of IL-4R interacts directly with rademikibart, which is absent in the dupilumab/IL-4R complex. This finding is confirmed by increased hydrogen bond interactions at the interface between rademikibart and IL-4R, demonstrating superior binding energy for rademikibart. Through analysis of the x-ray crystallography structures, MD-equilibrated structures, and computational point-mutation analysis of rademikibart, we identified residue Y50 and R55 of the light chain and R97, R99, and Y101 of the heavy chain of rademikibart as key residues interacting with IL-4R's third interface loop. Our data provides a molecular and structural rationale for the enhanced IL-4R inhibition by rademikibart over dupilumab, confirming rademikibart as an optimized second-generation IL-4R inhibitor.