NAADP elicits two-pore channel currents by lifting Lsm12-mediated inhibition of PI(3,5)P2 activation
NAADP elicits two-pore channel currents by lifting Lsm12-mediated inhibition of PI(3,5)P2 activation
Guan, X.; Du, C.; Shah, K. R.; Yan, J.
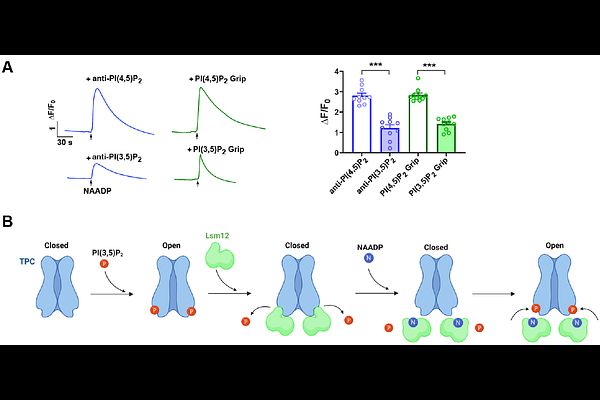
AbstractMammalian two-pore channels (TPCs) are endolysosomal cation channels that regulate membrane trafficking and ionic homeostasis and have been strongly implicated in nicotinic acid adenine dinucleotide phosphate (NAADP) signaling. We previously identified Lsm12 as an NAADP receptor and TPC-interacting protein required for NAADP-evoked Ca2+ mobilization from acidic organelles; however, how NAADP-Lsm12 coupling regulates TPC gating has remained unclear. Here, we show that Lsm12 acts as a potent antagonist of phosphatidylinositol 3,5-bisphosphate [PI(3,5)P2]-dependent TPC activation. Purified Lsm12 strongly inhibited PI(3,5)P2-evoked currents of TPC1 and TPC2, and endogenous Lsm12 similarly suppressed TPC2 activity in cells. Mechanistically, Lsm12 reduces the apparent sensitivity of TPC2 to PI(3,5)P2 through a competitive mechanism that depends on the concentrations of both Lsm12 and PI(3,5)P2, as well as on Lsm12-TPC interaction. Importantly, NAADP specifically and dose-dependently reverses Lsm12-mediated inhibition, restoring TPC currents only in the presence of PI(3,5)P2 or an intact PI(3,5)P2-binding site on TPCs. Consistently, acute sequestration of endogenous PI(3,5)P2 reduces NAADP-evoked cytosolic Ca2+ signals. These findings support a model in which Lsm12 tonically restrains PI(3,5)P2-dependent TPC gating, whereas NAADP binding to Lsm12 relieves this inhibition to permit channel activation. Our study therefore establishes a mechanistic link between NAADP signaling and phosphoinositide-dependent TPC gating and provides a working model for understanding NAADP-evoked Ca2+ release from acidic stores.