LOSS OF PARKIN DISRUPTS NUCLEAR AND MITOCHONDRIAL PROGRAMS REQUIRED FOR MUSCLE REGENERATION
LOSS OF PARKIN DISRUPTS NUCLEAR AND MITOCHONDRIAL PROGRAMS REQUIRED FOR MUSCLE REGENERATION
Gourlay, M.; Abbasi, M. R.; Cairns, G.; Thumiah-Mootoo, M.; Racine, J.; Ly, H. M.; Wang, A.; Larionov, N.; Blais, A.; Khacho, M.; Burelle, Y.
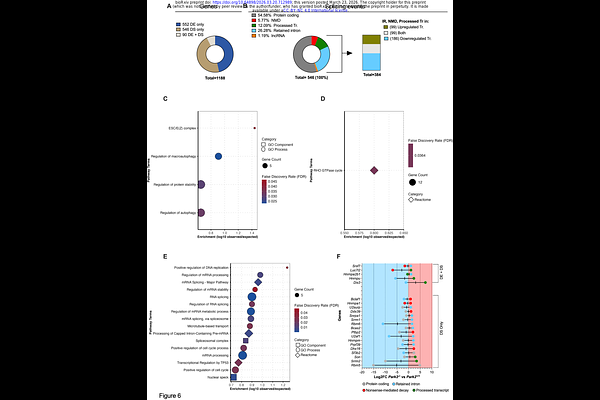
AbstractSkeletal muscle stem cells (MuSCs) rely on precisely coordinated metabolic and nuclear transitions to exit quiescence, enter the cell cycle, and regenerate tissue. How these processes are coupled remains poorly defined. Here, we identify PARKIN as a critical integrator of mitochondrial quality control and nuclear RNA processing programs that together enable balanced MuSC lineage progression. Using a MuSC-specific, inducible Park2 knockout model, we show that PARKIN supports mitophagy in quiescent MuSCs, and its loss triggers premature mitochondrial polarization and fragmentation - hallmarks of metabolic activation - that compromise appropriate self-renewal and fate specification. Unexpectedly, MuSCs harbor a constitutive nuclear pool of PARKIN that rises rapidly upon activation and localizes to interchromatin regions, with focal association with nuclear speckles. Park2-deficient MuSCs exhibit transcriptomic signatures consistent with widespread RNA isoform switching and intron retention, particularly affecting splicing machinery components, accompanied by altered nuclear speckle organization and impaired cell cycle progression. These findings reveal that PARKIN safeguards both mitochondrial homeostasis and the RNA processing architecture essential for activation, thereby coordinating metabolic and nuclear reprogramming during early MuSC state transitions. Our work positions PARKIN as a dual compartment regulator required for robust skeletal muscle regeneration.