Structural basis for saccharide binding by human RNase 2/EDN, a protein combining enzymatic and lectin properties
Structural basis for saccharide binding by human RNase 2/EDN, a protein combining enzymatic and lectin properties
Kang, X.; Prats-Ejarque, G.; Boix, E.; Li, J.
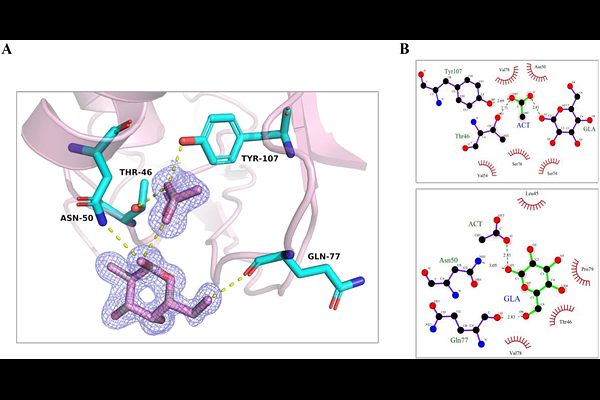
AbstractHuman RNase 2 (eosinophil-derived neurotoxin, EDN) is a major eosinophil granule protein of the vertebrate-specific RNase A superfamily and is involved in antiviral response and inflammation. Identifying ligand-binding pockets in EDN is thus relevant to structure-based drug design. In our laboratory we identified by protein crystallography a conserved site at the protein surface binding to carboxylic anion molecules (malonate, tartrate and citrate). Searching for potential biomolecules rich in anion groups and considering previous report of EDN binding to glycosaminoglycans, we explored the protein binding to saccharides. Next, EDN crystals were soaked with mono- and disaccharides, and the 3D structures of ten complexes were solved by X-ray crystallography at atomic resolution. We identified protein binding pockets to glucose, fucose, mannose, sucrose, galactose, trehalose, N-acetyl-D-glucosamine, N-acetylmuramic acid, and the sialic acid N-acetylneuraminic acid. A main site for glucose, fucose, and galactose was located adjacent to the spotted carboxylic anion site. Secondarily, N-acetylneuraminic acid, N-acetylmuramic acid, sucrose, galactose, and mannose shared another protein surface region. Overall, the saccharides clustered into seven defined sites, outlining a conserved recognition pattern, which was further analysed by molecular modelling. Interestingly, within the RNase A family, we find amphibian RNases that were initially isolated as carbohydrate binding proteins and named as leczymes, combining enzymatic and lectin properties. The present data is the first systematic structural characterization of a mammalian sugar-binding RNase within the family. The results highlight unique EDN residues that mediate its sugar specific interactions, of particular interest for a better understanding of the protein physiological role.