Ancient DNA reveals that natural selection has upregulated the immune system over the last 10,000 years
Ancient DNA reveals that natural selection has upregulated the immune system over the last 10,000 years
Maravall-Lopez, J.; Truong, B.; Kerner, G.; Zhao, Y.; Hou, K.; Perry, A.; Akbari, A.; Reich, D. E.; Price, A. L.
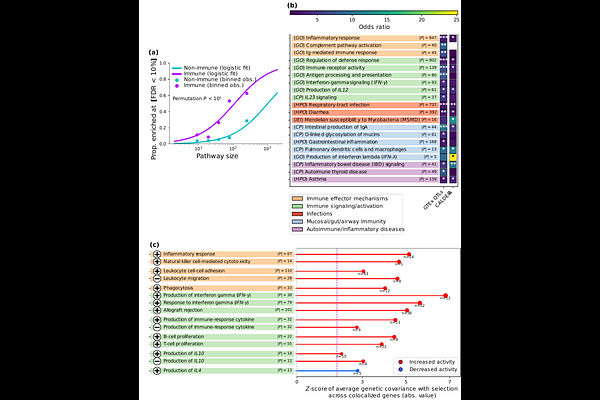
AbstractThe specific mechanisms through which human biology and disease susceptibility evolved with major shifts in West Eurasian environments and societies over the last 10,000 years --particularly rising infectious burden-- remain poorly characterized, despite ancient DNA studies identifying hundreds of candidate loci under positive selection. Here, we identify specific immune diseases/traits, genes/variants, pathways, and tissues/cell types impacted by natural selection by systematically integrating variant-level selection statistics with genome-wide association study (GWAS), quantitative trait locus (QTL), and molecular bulk/single-cell and gene pathway data. Genome-wide, positively-selected alleles are associated with reduced susceptibility to infectious diseases like tuberculosis (TB), influenza, and intestinal infections; consistent with selection-signal enrichments in immune cells within barrier tissues such as the respiratory tract and gut mucosa. In contrast, positively-selected alleles increase risk of intestinal inflammatory disease and autoimmune hypothyroidism, supportive of a tradeoff between infection and immune-mediated pathology, and consistent with adaptive alleles being QTLs for genes upregulating inflammation and other host-defense pathways. We reveal many novel adaptive loci with convergent signals from selection, infectious disease GWAS and immune-gene QTLs (including at FUT6 for intestinal infections; at ASAP1 for TB; and at LYZ, an antimicrobial enzyme), fine-mapping selection onto likely causal variants. Surprisingly, adaptive alleles had a protective effect on allergic conditions like asthma and dermatitis, challenging a common view that these conditions arose through evolutionary mismatch of present-day hygienic contexts relative to past, pathogen-rich environments.