Mueller glia-vasculature interactions in the developing retina
Mueller glia-vasculature interactions in the developing retina
Monshietehadi, S.; Garcia, A. J.; Smith, B. E.; Tarchick, M.; Conley, A.; Abhayaratne, K.; Feller, M.
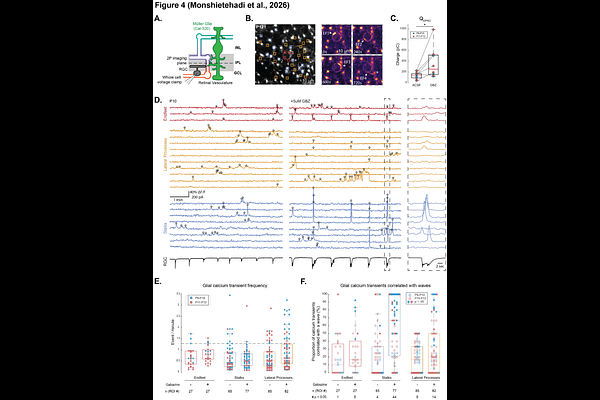
AbstractCoordinated signaling among neurons, glia, and the vasculature is essential for the formation of a functional nervous system, yet how these relationships emerge during development remains unclear. Here, we investigated the developmental interplay between neural activity, Mueller glia, and the retina vasculature in mice. Using quantitative confocal imaging from postnatal day 5 to eye-opening, we mapped the emergence of the superficial, intermediate, and deep vasculature layers and found that they emerged normally in mice lacking the beta2 containing nicotinic acetylcholine receptors, despite a dramatic reduction in cholinergic signaling. Tip cell density and overall vessel growth were unchanged, indicating cholinergic wave activity is not required for the emergence of retinal vasculature. We next defined the developmental timeline of Mueller glia-vascular interactions. Sparse labeling and immunohistochemistry revealed that Mueller glial lateral processes closely associate with endothelial tip cells during intermediate- and deep-layer angiogenesis and establish Aquaporin-4 enriched endfeet at vascular contact sites from the earliest stages of growth, even when vessel trajectories are perturbed. Finally, two-photon calcium imaging combined with simultaneous electrophysiology demonstrated that Mueller glial endfeet exhibit robust, compartmentalized calcium transients during development. Although a subset of events was temporally correlated with retinal waves, enhancing neurotransmitter spillover selectively increased wave-associated activity in glial stalks but not endfeet. These findings indicate that calcium signaling at the glial-vascular interface is largely independent of spontaneous neuronal activity. Together, our results support a model in which Mueller glia engage growing vessels through an activity-independent, parallel developmental program that may provide instructive cues for retinal angiogenesis.