Mechanotranscriptomic Profiling of Breast Cancer Cells Intravasated from Engineered Microtumors
Mechanotranscriptomic Profiling of Breast Cancer Cells Intravasated from Engineered Microtumors
Krueger, R.; Fuentes-Chandia, M.; Atiya, H.; De La Cruz, A.; Pashapour, S.; Boccaccini, A. R.; Selhuber-Unkel, C.; Kappelmann-Fenzl, M.; Bosserhoff, A.; Tobar, N.; Leal-Egana, A.
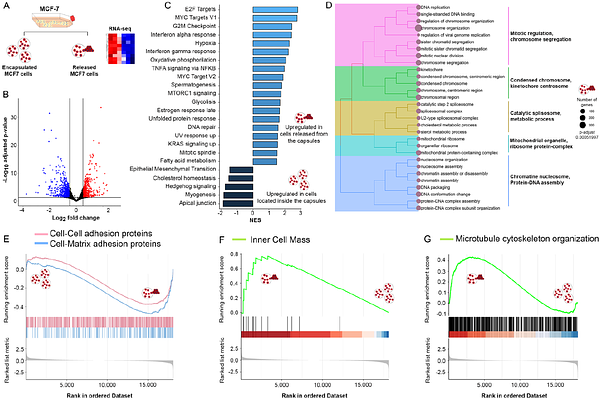
AbstractIntravasation is the process by which cancer cells breach the physical boundaries of a primary tumor and enter blood or lymphatic vessels. In this work, MCF-7 breast cancer cells were cultured within polymer-based microcapsules (here referred to as artificial microtumors) to investigate the transcriptomic and morpho-mechanical changes occurring in cancer cells during their release from these matrices, mimicking in vitro the process of intravasation. Our results show that even confined and released cancer cells share approximately 95% of their global transcriptomic profiles, intravasation-like cells exhibited marked differences in the expression of pathogenic hallmarks, including pathways associated with cell proliferation, immunosurveillance, and dormancy. Notably, a clear upregulation of YAP/TAZ signaling was observed in released cells, a result further supported by single-cell traction force microscopy assays, demonstrating that those cells exhibit higher biomechanical activity compared to cells located within artificial microtumors or those cultured on conventional 2D flasks, as shown for intravasated cells in vivo. To further enrich our investigation, the mechanotranscriptomic activity of MCF-7 cells was compared with suspended spheroids cultured on non-adherent surfaces (i.e., agarose hydrogels). Our results show that released cells displayed increased biomechanical activity and elevated expression of malignant markers, indicating that mechanical stress, beyond cell-cell contact alone, is required to trigger malignant responses. These observations were further supported by co-culture studies of MCF-7 cells with human fibroblasts and endothelial cells, which showed reduced proliferative and invasive capacities under confinement. Overall, our findings demonstrate that shifts in mechanical and metabolic stress, as experienced during intravasation, act as critical stimuli driving mechanotranscriptomic programs associated with cancer progression.