NaP-TRAP: A versatile and accessible workflow to dissect principles of translational regulation and mRNA stability
NaP-TRAP: A versatile and accessible workflow to dissect principles of translational regulation and mRNA stability
Gupta, A.; Struba, A. Z.; Madhavan, S.; Strayer, E.; Beaudoin, J.-D.
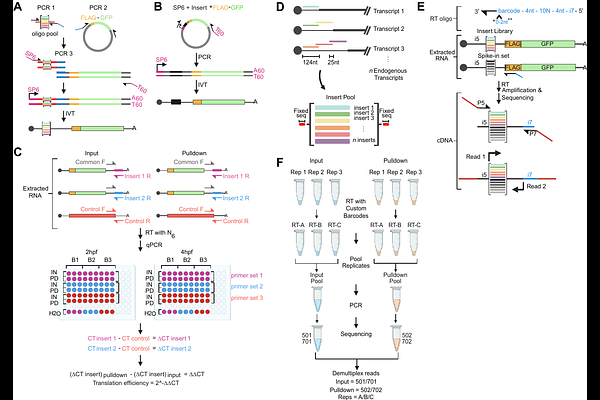
AbstractThe translation of mRNA into protein is tightly regulated by both cellular trans-factors and cis-regulatory elements encoded within transcripts. Although transcript fate can be measured by transcript abundance or translation efficiency, separating the contribution of each individual cis-element within a single transcript is an ongoing challenge. Current massively parallel reporter assay (MPRAs) approaches enable systematic interrogation of cis-regulatory elements that control transcript stability, but translation-focused MPRAs remain technically limited and often inaccessible. Here we present Nascent Peptide Translating Ribosome Affinity Purification (NaP-TRAP), a reporter-based approach that simultaneously measures translation and mRNA abundance. Unlike previous methods, NaP-TRAP captures translation directly through the immunoprecipitation of epitope-tagged nascent peptide chains, providing instantaneous, frame-specific readouts without specialized instrumentation. The method is highly scalable from single reporters to complex libraries, and adaptable across in vivo and in vitro systems. NaP-TRAP is versatile, allowing assessment of cis-regulatory impact of elements distributed throughout the mRNA, from cap-to-tail. This protocol covers experimental design, reporter construction, sample processing, and computational analysis for both low- and high-throughput applications. Bench work can be completed in 4-5 days, with qPCR-based readouts requiring only basic Excel skills for data processing. Sequencing-based readouts require skills in command-line tools and Python scripting and add an additional 2-3 days. NaP-TRAP thus offers an accessible, robust, and quantitative platform to decode the regulatory logic of mRNA translation and stability in diverse biological contexts.