Cross-species single-cell atlases chart progression, therapy-driven remodelling and immune evasion in pancreatic cancer
Cross-species single-cell atlases chart progression, therapy-driven remodelling and immune evasion in pancreatic cancer
Lucarelli, D.; Parikh, S.; Jimenez, S.; Schneeweis, C.; Ngandiri, D. A.; Putze, P.; Kos, T.; Wellappili, D.; Goelling, V.; Kuerbanjiang, M.; Shull, C.; Litwinski, M. R.; Handschuh, T. B.; Dabiri, Y.; Zukowska, M.; Seidler, B.; Kfuri-Rubens, R.; Baerthel, S.; Halle, L.; Arbesfeld-Qiu, J. M.; Gong, D.; Schneider, G.; Rad, R.; Falcomata, C.; Schmidt-Supprian, M.; Hwang, W. L.; Theis, F. J.; Saur, D.
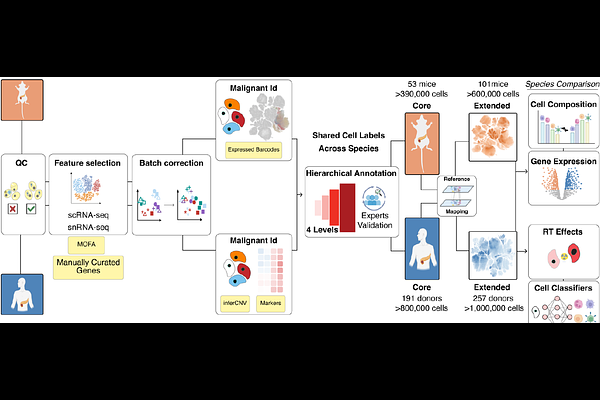
AbstractPancreatic ductal adenocarcinoma (PDAC) is typically diagnosed at advanced stages, yet single-cell datasets that capture late-stage and treated disease remain sparse, hindering progress in understanding tumour heterogeneity and therapy resistance. Here, we have generated integrated single-cell transcriptomic atlases of human and mouse PDAC to define the cellular and molecular landscape of the disease, from early to advanced and metastatic stages, including post-treatment disease, and to enable direct cross-species comparison. Using scANVI to harmonize 16 human studies comprising 257 donors and representative mouse models (101 tumours), we compiled over 1.6 million cells and established a four-level hierarchical taxonomy of more than 60 distinct cell states spanning malignant, stromal, immune, endothelial, adipose, exocrine and endocrine compartments. We resolve ten malignant programmes linked to progression and uncover rare immune phenotypes, including CD4CD8 double-positive T cells that remain poorly characterized in PDAC. Notably, we show that radiotherapy (RT) exposure is associated with enrichment of an EMT-persistent malignant state and an immunosuppressive microenvironment characterized by expansion of tumour-associated endothelium, depletion of intratumoral T cells and heightened laminin-CD44 signalling, with RT-associated genes linked to adverse prognosis in independent cohorts. Cross-species mapping reveals that orthotopic syngeneic allografts more faithfully recapitulate the cellular diversity and EMT-enriched states of advanced human PDAC, underrepresented in autochthonous genetically engineered models, with differences driven primarily by cell-type composition rather than pathway divergence. Together, these atlases and pretrained models provide a broadly accessible reference for benchmarking PDAC model fidelity and for interrogating mechanisms of tumour progression, microenvironmental remodelling and therapy response and resistance.