Modulation of Oncogenic KRAS Signaling by Branched Actin-driven Cell Membrane Protrusions
Modulation of Oncogenic KRAS Signaling by Branched Actin-driven Cell Membrane Protrusions
Gihana, G. M.; Bhatt, K.; Siruvallur Murali, V.; Zhou, F. Y.; Noh, J.; Ravishankar, R.; Borges, H.; Lin, J.; Oceguera, J.; Nogueira, P. A. S.; Perez-Castro, L.; Venkateswaran, N.; Chen, B.; Fiolka, R. P.; Conacci-Sorrell, M.; Dean, K. M.; Danuser, G.
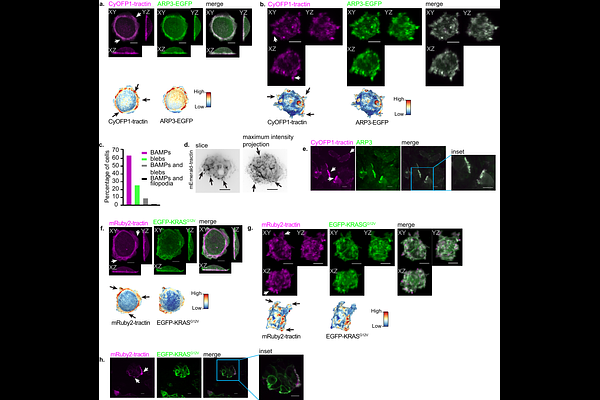
AbstractFor over three decades, we have known that oncogenic RAS alters the actin cytoskeleton organization and cell surface morphology (1,2). RAS activates the GTPase RAC1, which triggers the growth of branched actin networks to promote cell membrane protrusions (3,4). In melanoma, the hyperactive RAC1 mutant, Rac1P29S, was recently shown to drive extended lamellipodia, which then empower cell proliferation through sequestration and localized inhibition of the merlin tumor suppressor (5). This discovery illustrates cell morphological programs not only as outputs but also as regulators of human oncogenic signals. Hence, we wondered whether the pronounced branched actin-driven membrane protrusions (BAMPs) downstream of oncogenic RAS are not mere outputs of RAS signaling but rather an active component in mediating the oncogenic penetrance of RAS mutants. We used volumetric light sheet microscopy and biochemical approaches to investigate the role of BAMPs in regulating the molecular signaling of oncogenic KRAS in pancreatic and lung cancer models. We found that elevated BAMP formation regulated the interaction of oncogenic KRAS with downstream effectors, specifically with the RAC1 GEF TIAM1. This implies that BAMPs amplify their own upstream regulators in a positive feedback. This meritorious cycle upregulates cyclin D1 expression by inactivating the merlin tumor suppressor, independently of the mitogen activated protein kinase pathway (MAPK). In the absence of BAMPs, cells carrying oncogenic KRAS mutations are unable to attain their full penetrance in proliferation. Overall, this work unveils the long-overlooked role of branched actin-driven cell morphology in the functionalization of KRAS mutants as potent oncogenes.