Matrix metalloproteinases proteolyze RAB proteins and contribute to cisplatin-induced ototoxicity
Matrix metalloproteinases proteolyze RAB proteins and contribute to cisplatin-induced ototoxicity
Bhavsar, A. P.; Zandi, Z.; Hartley, B.; Bassiouni, W.; DuVal, M. G.; Luo, S.; Spavor, M. J.; Allison, W. T.; Julien, O.; Schulz, R.
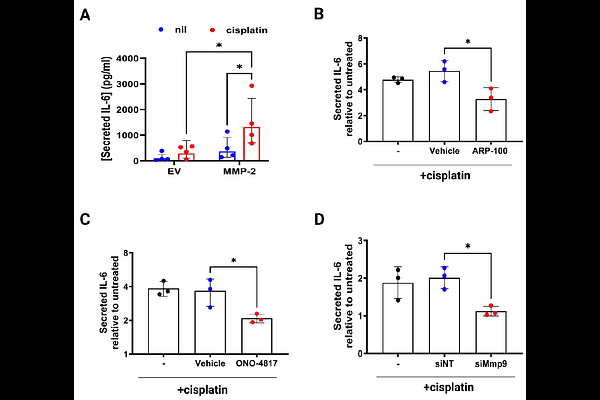
AbstractMatrix metalloproteinases (MMPs) are rapidly expressed and activated in response to oxidative stress and contribute to various pathological conditions. Cisplatin is a highly effective chemotherapeutic agent; however, its clinical use is limited by its associated permanent hearing loss (ototoxicity). While cisplatin-induced oxidative stress and inner ear cell death are well-established, the contribution of MMPs remains unclear. In this study, we demonstrate that cisplatin exposure triggers activation of MMP-2 and MMP-9 and expression of an intracellular N-terminal-truncated isoform of MMP-2 in mouse inner ear hair cells. Pharmacological inhibition of MMP-2 and genetic knockdown of Mmp-9 enhanced hair cell survival and attenuated cisplatin-induced inflammation and cytotoxicity. Furthermore, proteomic analysis revealed that proteins involved in intracellular trafficking, including RAB proteins, may serve as potential substrates of intracellular MMP-2 upon cisplatin exposure, pointing to a previously unrecognized mechanism of cisplatin-induced hair cell injury. In vitro analysis confirmed that MMP-2 cleaves RAB9A in response to cisplatin, and in silico analyses predicted MMP-2-preferred cleavage sites on RAB9A. Collectively, our findings identify MMP-2 as a promising therapeutic target for mitigating cisplatin-induced ototoxicity.