Organoids serve as viable in vitro model for functional precision medicine for mesonephric-like adenocarcinoma of the ovary
Organoids serve as viable in vitro model for functional precision medicine for mesonephric-like adenocarcinoma of the ovary
Holthaus, D.; Le, H. D.; Matzner, L.; Kellers, F.; Rogmans, C.; Winkler, V.; Bastian, L.; Fliedner, S.; Weimer, J. P.; Busch, H.; Mandelkow, T.; Konukiewitz, B.; Maass, N.; van Mackelenbergh, M.; Alkatout, I.; Bauerschlag, D. O.; Hedemann, N.
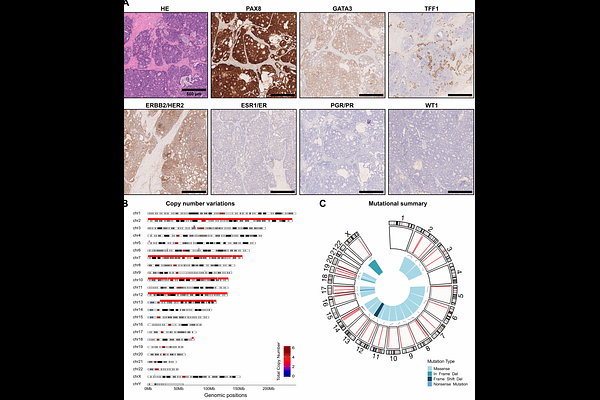
AbstractBackground: Mesonephric-like adenocarcinoma has been recently classified as a rare type of ovarian carcinoma. Description of these tumours have been rare and mostly covered in case reports. In some cases, molecular characterization by sequencing has been employed for guided therapy recommendations, however, functional chemosensitivity testing of targetable pathways using advanced in vitro cellular models such as organoids has not been reported so far. Here, we report on a case of ovarian cancer that was later identified as mesonephric-like adenocarcinoma at an advanced stage. Methods: The tumour was characterized by molecular techniques including immunohistochemistry and whole-exome sequencing. At the same time, ovarian cancer organoids were established by adapting existing protocols for high-grade serous ovarian carcinoma. The organoids were subsequently used for functional in vitro chemosensitivity testing by treatment with standard-of-care chemotherapeutics cisplatin, paclitaxel, and the Poly (ADP-Ribose) Polymerase 1-inhibitor olaparib. Based on molecular characteristics, we also applied the inhibitor binimetinib, to target Mitogen-Activated Protein Kinase downstream of the KRAS Proto-Oncogene. Additionally, chemotoxicity testing with healthy fallopian tube organoids and high-grade ovarian cancer organoids was applied to determine the therapeutic window. Results: Immunohistochemical analysis showed characteristic PAX8+, GATA3+, TFF1+, ER-, PR-, WT1- staining while the sequencing revealed mutations in 31 genes of which KRAS G12V and DYNC1H1 G4072S were annotated as (likely) pathogenic. The tumour was mismatch-repair proficient. Tumour-derived organoids proved to be highly resistant to standard-of-care chemotherapeutics cisplatin, paclitaxel, and olaparib, but sensitive to inhibition by binimetinib, which aligned well with the molecular characteristics. Direct comparison to healthy fallopian tube organoids and high-grade ovarian cancer organoids confirmed low cytotoxic potential underlining a feasible therapeutic window for binimetinib. Conclusions: For the first time, we show that existing protocols for high-grade serous ovarian carcinoma can be used for the generation of organoids derived from mesonephric-like adenocarcinoma. These organoids could be used as an essential tool for functional precision medicine purposes. This functional data could be applied as an additional layer for molecular tumour boards diagnostics by supporting molecular datasets and even identify targetable pathways beyond genetic variations, thus offering novel therapeutic options particularly for rare and aggressive tumours.