Stereospecific rapid activation of Transcription Factor EB (TFEB) by Levacetylleucine (NALL)
Stereospecific rapid activation of Transcription Factor EB (TFEB) by Levacetylleucine (NALL)
Galione, A.; Platt, F. M.; Churchill, G. C.; Patterson, M. C.; Factor, M.; Fields, T.; Davis, L. C.; Strupp, M.
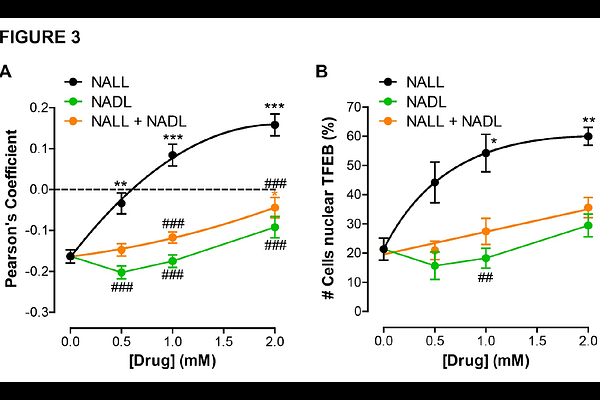
AbstractN-acetyl L-leucine (NALL, USAN or levacetylleucine, INN or trade name Aqneursa) is an FDA-approved agent for the treatment of Niemann-Pick disease type C (NPC). The N-acetyl group renders the compound a prodrug of L-leucine, making it a substrate for membrane-spanning monocarboxylate transporters (MCTs), which are ubiquitously expressed delivering NALL to all tissues with high capacity, including to the central nervous system. NALL enters enzyme-controlled pathways that correct metabolic dysfunction and enhance mitochondrial bioenergetics. Because NALL improves energy production (adenosine triphosphate, ATP) and ameliorates lysosomal function, it is potentially a therapy for a broad range of neurodegenerative and neurodevelopmental disorders (in addition to lysosomal storage disorders) in which energy homeostasis and lysosomal function are impaired. Here, we have performed a series of in vitro studies which reveal an additional aspect of NALLs polypharmacological mechanism of action. The studies demonstrate a direct lysosomal effect whereby NALL rapidly activates the translocation of the Transcription Factor EB (TFEB, a master regulator of lysosomal biogenesis and autophagy) from the cytoplasm to the nucleus in HeLa cells. The activation of TFEB is known to trigger the activation of specific genes that restore lysosomal biogenesis and function, as well as autophagy. Consistent with this, we show that NALL increases production of a TFEB target gene LAMP1, an integral lysosomal membrane protein responsible for maintaining lysosomal integrity, function and pH. This in vitro effect occurs at concentrations consistent with concentrations in plasma in humans after standard therapeutic dosing. We further demonstrated that acetylation is critical to this aspect of NALLs mechanism of action, as L-leucine itself had no effect on the activation of TFEB. Consistent with previous studies N-acetyl-D-leucine was inactive and also had no effect. Similarly, N-acetyl-DL-leucine also had only a modest effect, providing further evidence that N-acetyl-D-leucine is even antagonistic and inhibits the effects of the active L-enantiomer. This mechanism of action of NALL to activate TFEB signalling, thereby enhancing lysosomal and autophagic function, further elucidates the ways by which this compound targets the fundamental etiology of rare and common neurodegenerative disorders, from Niemann-Pick disease type C to Parkinsons disease. Based on its mechanism of action by improving the mitochondrial-lysosomal axis, NALL has the potential to be an effective therapy for a broad range of neurological and neurodevelopment conditions.