Thirty years of Achromobacter ruhlandii evolution reveal pathways to epidemic lineages
Thirty years of Achromobacter ruhlandii evolution reveal pathways to epidemic lineages
Gabrielaite, M.; Johansen, H. K.; Juozapaitis, J.; Marvig, R. L.; Dudas, G.
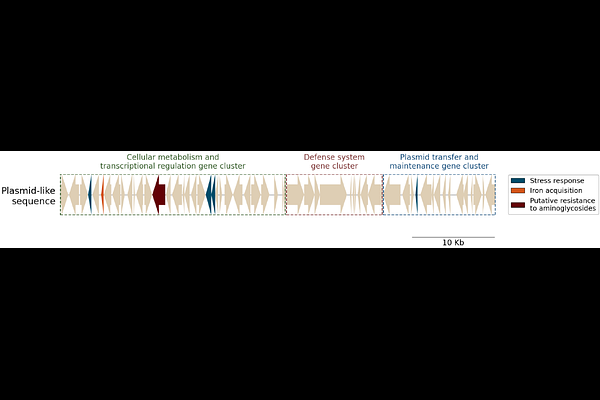
AbstractBackground: Achromobacter spp. are emerging opportunistic pathogens, associated with chronic infections, antimicrobial resistance, and poor clinical outcomes. The Danish epidemic strain (DES) of A. ruhlandii is highly drug-resistant and adapted to the cystic fibrosis (CF) airway, yet its evolutionary history and defining genomic features remain poorly understood. Methods: We analysed genome and antibiotic susceptibility testing data for 58 longitudinally collected DES isolates sampled over 21 years at Rigshospitalet, Denmark. We combined these with 79 publicly available A. ruhlandii genomes and applied phylogenomics to infer DES emergence and transmission, and genome-wide association studies (GWAS) to identify lineage-specific and adaptive genomic features. Results: DES forms a distinct monophyletic clade within A. ruhlandii, estimated to have emerged around 1990, with no evidence of dissemination beyond Denmark. GWAS identified key lineage-defining traits, including acquisition of large mobile genetic elements, plasmid integration events, and enrichment of resistance and iron acquisition genes. In addition, we detected other epidemic A. ruhlandii lineages with evidence of long-term persistence and inter-country spread, sharing similar genetic signatures of adaptation. Conclusions: This study elucidates the genomic features associated with chronic infection and epidemic potential in A. ruhlandii. The DES lineage illustrates how extensive horizontal gene transfer, high intrinsic resistance potential, and enhanced host-adaptation traits, such as increased iron acquisition, can facilitate the emergence and persistence of successful epidemic lineages. These findings highlight shared evolutionary signatures of epidemic A. ruhlandii and underscore the need for continued genomic surveillance to detect and monitor emerging high-risk lineages in chronic infections.