In situ cryo-ET of mammalian embryos reveals cytoplasmic lattices contain ubiquitin-charged E2-E3 ligase assemblies
In situ cryo-ET of mammalian embryos reveals cytoplasmic lattices contain ubiquitin-charged E2-E3 ligase assemblies
Singh, K.; Harasimov, K.; Niakan, K. K.; Carter, A. P.
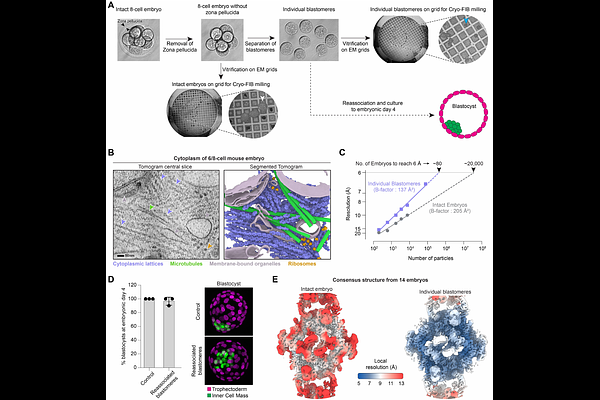
AbstractCytoplasmic lattices (CPLs) are filamentous assemblies essential for mammalian embryonic development that regulate organelle organization, spindle assembly, and protein homeostasis. However, their molecular functions remain unclear. Here, we develop a strategy combining cryo-focused ion beam milling and cryo-electron tomography to resolve macromolecular complexes directly in mammalian embryos. Using this approach, we determine the in situ structure of CPLs within mouse embryos at ~4.7 [A] resolution. CPL filaments are built from multiple copies of fourteen proteins arranged into a ~4.5 MDa repeating unit. These components form a scaffold that organizes a central cavity containing three complexes of the E2 ubiquitin-conjugating enzyme UBE2D, the E3 ligase UHRF1, NLRP14 and {beta}-tubulin. We observe states in which UBE2D is conjugated to ubiquitin, along with structural rearrangements within the cavity consistent with regulated ubiquitin transfer. Together, our findings suggest that CPLs are large ubiquitin ligase assemblies and play a role in post-translational protein modification during early embryonic development.