Suppression of early pro-inflammatory senescent signature post-radiotherapy mitigates chronic bone damage
Suppression of early pro-inflammatory senescent signature post-radiotherapy mitigates chronic bone damage
Achudhan, D.; Orme, J.; Sharma, R.; Komel, A.; Khan, K. G.; White, T. A.; LeBrasseur, N. k.; Khosla, S.; Park, S. S.; Pignolo, R. J.; Chandra, A.
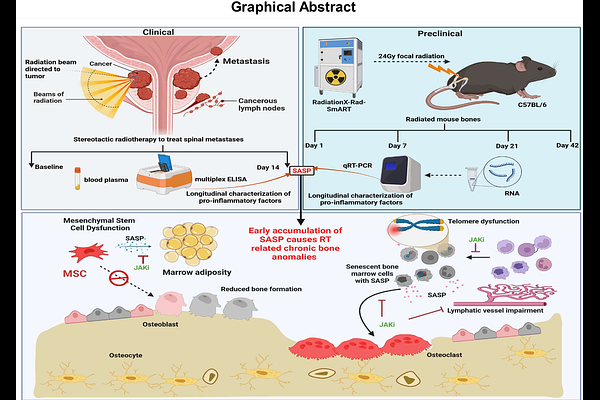
AbstractCellular senescence has been implicated in the pathophysiology of radiotherapy-related bone loss. Based on our previous work, clearance of senescent cells using genetic and pharmacological tools alleviates the anomalies associated with radiation-associated bone deterioration. The pro-inflammatory senescence associated secretome referred to as senescence associated secretory phenotype (SASP), is a hallmark of cellular senescence. The modulation of SASP by senomorphic drugs, potentially can suppress the pro-inflammatory secretome of senescent cells, irrespective of the underlying senescence mechanism. In this study we tested a senomorphic drug, ruxolitinib, a Janus kinase inhibitor (JAKi), during acute and chronic radiotherapy related effects on the bone. Our clinical data indicate an early increase in several pro-inflammatory SASP proteins following radiotherapy of spinal metastasis in prostate cancer patients. Longitudinal assessment of SASP-related genes confirmed this acute elevation in several SASP markers in systemic circulation following irradiation of mouse femurs. In a proof-of-concept study, following two preclinical radiotherapy regimens of cumulative doses of 30Gy (5 x 6Gy) and 60Gy (5 x 12Gy), a senomorphic approach of JAKi treatment was more effective in alleviating radiation-related bone loss compared to the senolytic cocktail of D+Q. Early and intermittent suppression of SASP using JAK inhibitors alleviated chronic bone deterioration, diminished telomere dysfunction, lowered senescence and SASP marker expression, reduced bone marrow adiposity, and mitigated radiation related lymphatic impairment. Overall, our study shows that early targeting of SASP proteins could be a potential therapeutic to prevent radiotherapy-related chronic bone loss and risk of fractures.